최소 단어 이상 선택하여야 합니다.
최대 10 단어까지만 선택 가능합니다.
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
NTIS 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
DataON 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Edison 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Kafe 바로가기
| 주관연구기관 | 건국대학교 KonKuk University |
|---|---|
| 보고서유형 | 최종보고서 |
| 발행국가 | 대한민국 |
| 언어 | 한국어 |
| 발행년월 | 2015-02 |
| 과제시작연도 | 2013 |
| 주관부처 | 농촌진흥청 Rural Development Administration(RDA) |
| 과제관리전문기관 | 농촌진흥청 Rural Development Administration |
| 등록번호 | TRKO201500010656 |
| 과제고유번호 | 1395031344 |
| 사업명 | 차세대바이오그린21 |
| DB 구축일자 | 2015-07-11 |
| DOI | https://doi.org/10.23000/TRKO201500010656 |
Ⅳ. 연구개발결과
■ 대장균을 이용하여 포도당으로 부터 caffeic acid를 생산함. Caffeic acid의 생산량을 늘리기 위해 caffeic acid의 전구물질인 tyrosine의 합성을 증대 시킨 대장균 균주를 이용함.
■ Caffeic acid생산 균주의 반응물을 이용하여 두 번째 대장균 균주를 이용하여 chlorogenic acid를 생산함. 생산량은 78 mg/L였음.
■ Hydroxycinnamic acid-glycerol기반 chiral compound를 대장균을 이용하여 만듬.
■ Hydo
Ⅳ. 연구개발결과
■ 대장균을 이용하여 포도당으로 부터 caffeic acid를 생산함. Caffeic acid의 생산량을 늘리기 위해 caffeic acid의 전구물질인 tyrosine의 합성을 증대 시킨 대장균 균주를 이용함.
■ Caffeic acid생산 균주의 반응물을 이용하여 두 번째 대장균 균주를 이용하여 chlorogenic acid를 생산함. 생산량은 78 mg/L였음.
■ Hydroxycinnamic acid-glycerol기반 chiral compound를 대장균을 이용하여 만듬.
■ Hydorxycinnamic acid-glycerol의 생산량은 약 1000 mg/L이상이었음. p-Coumaroyl glycerol의 입체순도는 약 69%였음.
■ 합성생물학 기반 발현벡터인 piBR181 발현벡터 개발
■ 대장균에서 생합성 경로 재설계와 합성생물학 기반에 의한 당합성 및 당전이 균주 개발
■ YjiC 당전이효소를 이용한 Phloretin 반응으로부터 5개의 phloretin glycoside 화합물 얻었음. 그중에 3개의 화합물은 신물질이며, 구조 결정 및 생리활성 확인
■ YjiC 당전이효소를 이용하여 resveratrol에 당전이 반응하여 총 4개의 resveratro glycoside를 합성 하였으며, 구조 결정 및 생리활성
■ 7-hydroxy-8-methoxyflavone 에 glucose. galactose 및 2-deoxyglucose 신규물질 합성
■ Genistein 4′,7-O-β-D-diglucoside, Sissotrin, daidzein 4′-O-β-D-glucoside 및 daidzin 합성
■ 4′,7-O-β-D-diglucoside, and formononetin 7-O-β-D-glucoside, a-mangostin glucoside alc 2-deoxyglucoside합성 및 구조 결정
■ 스타틴계 유도체 개발에 필요한 유용 cytochrome P450 유전자 활용 시스템 개발: P450 효소의 활성 측정기술 개발, HTS system 개발, chimeric mutants의 제조
■ 스타틴계 화합물(Simvastatin, Lovastatin, Atorvastatin, Fluvastatin, Monacolin J)로부터 hydroxylated statin derivatives (대사체) 제조 기술 및 분석기술 개발
■ 스타틴계 유도체 개발에 필요한 유용 유전자 개발 및 활용기술 개발
■ 스타틴계 약물의 대사체 효능검증 기술 개발: in vitro 및 in vivo system
■ 스타틴계 약물의 대사체로부터 HMG-CoA reductase 저해제 개발
■ 스테로이드계 화합물 17beta-Estradiol의 대사체 및 유도체 개발에 활용 가능함
■ 본 연구에서 개발한 P450 효소를 활용하여 임상적으로 사용되는 다양한 약물의 대사체 제조에의 응용성을 확인함: Diclofenac, tamoxifen, propafenone, astermizole
■ Chromogenic 및 fluorogenic substrates 활용 Chimeric mutants의 활성 screening 방법 개발: p-Nitrophenol hydroxylation, 7-ethoxycoumarin O-deethylation
■ 이종숙주 미생물 대장균 내 핵산당 고체상 추출법 확립
■ 핵산당 표준품을 활용한 고감도 검출 HPLC-ESI-MS법 구축
■ 이종숙주 E. coli의 배양시간 별 샘플링을 통한 핵산당 프로파일 모니터링
■ 관련 생합성 유전자 과발현을 통한 재조합 대장균 내 핵산당 플럭스의 변화 분석 및 플랫폼 설계
■ 플라보노이드 및 당유도체의 항비만 및 지질대사 개선 연구: Quercetin, Quercetin-3-glucoside, 7'8'-Dihydroxyflavone를 이용하여 지방세포에 대한 세포독성과 지방세포 내 지방축적 억제 효과를 확인
■ 플라보노이드 및 당유도체의 면역증강 효과 연구: 내츄럴 플라보노이드 보다 플라보노이드의 당 유도체들이 면역세포의 생존력을 더 증가시킴으로서 면역증강효과가 더 우수함을 확인 하였다.
■ 플라보노이드 및 당유도체의 항염증 및 항알레르기성 천식에 대한 효능 연구: phloretin과 biochanin A (BioA)이 항알레르기성 항천식 효과를 확인. kaempferol과 kaempferol-3-O-rhamnoside의 세포레벨 및 천식 동물모델에서의 항천식 효과를 규명
1. Hydroxycinnamic acid-quinate is major dietary phenolic as well as antioxidant, with recently discovered biological activities including protection against chemotheraphy side effects and prevention of cardiovascular disease and cancer. Certain fruits and vegetables produce these compounds, though
1. Hydroxycinnamic acid-quinate is major dietary phenolic as well as antioxidant, with recently discovered biological activities including protection against chemotheraphy side effects and prevention of cardiovascular disease and cancer. Certain fruits and vegetables produce these compounds, though a microbial system can also be utilized for synthesis of hydroxycinnamic acid-quinate. In this study, we engineered Escherichia coli to produce chlorogenic acid from glucose. For the synthesis of chlorogenic acid, two E. coli strains were used; one strain for the synthesis of caffeic acid from glucose and the other strain for the synthesis of chlorogenic acid from caffeic acid and quinic acid. The final yield of chlorogenic acid using this approach was approximately 78 mg/L. Hydroxycinnamoyl glycerol can be used as a chemical synthon to synthesize chiral compound. Hyroxycinnamoyl glycerol was synthesized in E. coli and the enotiomeric specificity of the hydroxinnamoyl glycerol was determined.
2. To produce naturally metabolites, the biosynthetic genes must be arranged with a high efficient cloning strategy capable of manifesting a new biological environment. A simple and commercially available bio-part, despite the current trend focusing on the standardized BioBricks bio-parts available in the registry is used. A 181-bp nucleotide fragment was designed for the efficient construction of an expression vector for monocistronic assembly of genes. Expression vector is pIBR181.
A multi-monocistronic synthetic vector system (pIBR181) was employed to construct robust genetic circuits for the production of valuable flavonoid glycosides in Escherichia coli. Characterized functional genes involved in the biosynthesis of uridine diphosphate (UDP)-glucose and thymidine diphosphate (TDP)-rhamnose from various microbial sources along with glucose facilitator diffusion protein (glf) and glucokinase (glk) from Zymomonas mobilis were assembled and overexpressed in a single synthetic multi-monocistronic vector. The newly generated nucleotide diphosphate (NDP)-sugars biosynthesis circuits along with regiospecific glycosyltransferases (GTs) from plants were introduced in E. coli BL21 (DE3) to probe the bioconversion of fisetin, a medicinally important polyphenol produced by various plants. As a result out of 0.9 g fisetin, approximately 1.178 g of fisetin 3-O-glucoside and 1.026 g of fisetin 3-O-rhamnoside were produced in UDP-glucose and TDP-rhamnose biosynthesis systems respectively, after 48 h of incubation in 3 L fermentor. The systems were found to be highly effective in bio-transforming other flavonols (quercetin, kaempferol, myricetin) into their respective glycosides, achieving over 95% substrate conversion.
A UDP-glycosyltransferases from Bacillus licheniformis was exploited for the glycosylation of phloretin. The in vitro glycosylation reaction has confirmed the production of five phloretin glucosides, including three novel glucosides. Consequently, we demonstrated the application of the same glycosyltransferase for the efficient whole-cell biocatalysis of phloretin in engineered E. coli . Phloretin 4’, 4-O-diglucoside, Phloretin 4,6’-O-diglucoside and Phloretin 2’,4’,4-O-triglucoside have not been reported from a natural source.
Also YjiC was incubated with nucleotide diphosphate (NDP) D- and L-sugars to produce glucose, galactose, 2-deoxyglucose, viosamine, rhamnose, and fucose sugar-conjugated resveratrol glycosides. A significantly higher (90%) bioconversion of resveratrol was achieved with UDP-D-glucose as the sugar donor, while the conversion rates and number of products formed were found to vary with the other NDP-sugar donors. These results confirmed the ability of YjiC to accept diverse donor substrates. In cell viability assays of macrophage cells, two different resveratrol monoglucosides, (E)-resveratrol 3-O-β-D-glucoside and (E)-resveratrol 4ʹ-O-β-D-glucoside, exhibited significantly reduced cytotoxicity.
YjiC has been used for the modification of the commercially available isoflavonoids genistein, daidzein, biochanin A and formononetin. The in vitro glycosylation reaction, using UDP-α-D-glucose as a donor for the glucose moiety and aforementioned four acceptor molecules, showed the prominent glycosylation at 4' and 7 hydroxyl groups, but not at the 5thhydroxylgroupoftheA-ring,resultingintheproductionofgenistein4'-O-β-D-glucoside, genistein 7-O-β-D-glucoside (genistin), genistein 4',7-O-β-D-diglucoside, biochanin A-7-O-β-D-glucoside (sissotrin), daidzein 4'-O-β-D-glucoside, daidzein 7-O-β-D-glucoside (daidzin), daidzein 4', 7-O-β-D-diglucoside, and formononetin 7-O-β-D-glucoside (ononin). The structures of all the products were elucidated using high performance liquid chromatography-photo diode array and high resolution QTOF-ESI/MS analysis, and were compared with commercially available standard compounds. Significantly higher bioconversion rates of all four isoflavonoids was observed in both in vitro as well as in vivo bioconversion reactions. The in vivo fermentation of the isoflavonoids by applying engineered E. coli BL21(DE3)/Δpgi ΔzwfΔushA overexpressing phosphoglucomutase (pgm) and glucose 1-phosphate uridyltransferase (galU), along with YjiC, found more than 60% average conversion of 200 μM of supplemented isoflavonoids, without any additional UDP-α-D-glucose added in fermentation medium, which could be very beneficial to large scale industrial production of isoflavonoid glucosides.
Two enzymatic systems were developed for efficient synthesis of glycosides of a-mangostin, a natural xanthonoid exhibiting antioxidant, anti-bacterial, anti-inflammatory, and anti-cancer activities. In these systems, one-pot reactions for synthesis of UDP-α-D-glucose and TDP-α-D-2-deoxyglucose were modified and combined with a glycosyltransferase (GT) from Bacillus licheniformis to produce C-3 and C-6 positions modified glucose as well as 2-deoxyglucose conjugated novel α-mangostin derivatives. As a result, a-mangostin 3-O-β-D-glucopyranoside, a-mangostin 6-O-β-D-glucopyranoside, a-mangostin 3,6-di-O-β-D-glucopyranoside, a-mangostin 3-O-β-D-2-deoxyglucopyranoside, a-mangostin 6-O-β-D-2-deoxyglucopyranoside, and a-mangostin 3,6-di-O-β-D-2-deoxyglucopyranoside were successfully produced in practical quantities and characterized by high resolution quadruple-time of flight-electrospray ionization-mass spectrometry (HR-QTOF ESI/MS) and 1H-and13C-NMRanalyses. In excess of substrate, the maximum productions of three a-mangostin glucopyranosides (4.8 mg/mL, 86.5% overall conversion of a-mangostin) and of three a-mangostin 2-deoxyglucopyronosides (4.0 mg/mL, 79% overall conversion of a-mangostin) were achieved at 4 h reaction incubation. All the α-mangostin glycosides exhibited improved water-solubility whereas the antibacterial activity of a-mangostin against three Gram-positive bacteria Micrococcus luteus, Bacillus subtilis, and Staphylococcus aureus was drastically enhanced by glucosylation at C-3 position. This study replaced the use of expensive NDP-sugars in the glycosylation reactions and offers a scheme that could be useful to produce diverse glycosylated natural products in significant quantities by using inexpensive starting materials and recycling co-factors within a reaction vessel.
3. One of Goals of our research was "Development of Drug Leads for Hyperlipidemia
Using Bacterial Metabolic Engineering." To achieve the goal, the following specific aims were performed: molecular evolution of bacterial P450 enzyme genomics, development of HTS system for P450 activities and expression, generation of engineered enzymes, whole-cell biocatalysis, design and generation of human drug metabolites and their derivatives. To achieve the specific aims, molecular evolution of bacterial P450 genomics and developments of HTS systems for P450 expression, catalytic activities, and ligand binding were performed. To set up a general system for the drug and lead diversification with human statin metabolites, analysis and separation of metabolites of drugs and lead compounds were performed. For the generation and analysis of combinational chemical library, statin drugs such as atorvastatin, simvastatin, fluvastatin and lovastatin were used. For the analysis of metabolite derivatives, mass production and separation of drug metabolites and lead compounds were done. For this specific aim, engineering bacterial P450 enzymes were developed by using domain swapping of heme and reductase domain and directed evolution of heme domains of highly active chimera. HTS systems for the screening of highly active chimeric mutants, chromogenic and fluorogenic substrates were found. In this study we were trying to do generation of human metabolites of statin drugs at first and to make several derivatives of those statin metabolites. The target for our study includes atorvastatin, simvastatin, fluvastatin and lovastatin. Production of derivatives of the metabolites resulted in generation of chemical library of statin derivatives. We developed HTS systems for the evaluation of statin chemicals on the inhibitory effect on the HMG CoA reductase activity. Inhibitory effect of the statin derivatives on the HMG-CoA reductase were examined. All of the hydroxylated statin derivatives showed inhibitory effect on that enzyme activity, this could be possible candidates for the hyperlipidemia. The inhibitory effects were comparable to the parent statin drugs. For those with in vitro activity, in vivo experiments with animal model were also performed to confirm the effect of statin derivatives on the animal model.
Present results should be useful to satisfy the demand of the revised Food and Drug Administration (FDA) guidelines for evaluating drug toxicity, particularly with regard to the toxicity of drug metabolites. In February 2008, the FDA issued the Guidance for Industry:Safety Testing of Drug Metabolites. According to this guidance, any human drug metabolites“. formed at greater than 10 percent of parent drug systemic exposure at steady state” should be subject to separate safety testing, that is, by synthesis and administration to test animals The issue of human metabolites in safety testing (MIST) has presented a challenge at the early stages of drug development for the pharmaceutical industry. Bacterial P450 BM3 mutants can be used to produce the metabolites of human P450-catalyzed reactions. We found other advantages of using mutants of P450 BM3 (CYP102A1) from Bacillus megaterium instead of human P450 enzymes.
4. The primary aim of “omic” technologies is the non-targeted identification of all gene products (transcripts, proteins, and metabolites) present in a specific biological sample. By their nature, these technologies reveal unexpected properties of biological systems. A second and more challenging aspect of omic technologies is the refined analysis of quantitative dynamics in biological systems. For metabolomics, liquid chromatography coupled to mass spectrometry are well suited for coping with high sample numbers in reliable measurement times with respect to both technical accuracy and the identification and quantitation of small-molecular-weight metabolites. This potential is a prerequisite for the analysis of dynamic systems. Thus, metabolomics is a key technology for systems biology. The aim of their analyses in microbial biotechnology is to provide comprehensive insight into the metabolic state of the target microbe by detecting the metabolome - the full suite of metabolites expressed in a host.
There is a long tradition of, and extensive knowledge about, metabolite analysis. In fact, metabolite analysis can be better understood by distinguishing among levels on the basis of its objectives. First, there is metabolite target analysis, which utilizes specialized protocols for the analysis of different analytes. Second, metabolite profiling aims at quantitation of several pre-defined targets. Third, metabolomics has the ultimate goal of unbiased identification and quantitation of all the metabolites present in a certain biological sample from an organism grown under defined conditions. Among these three approaches, metabolomics seems to be best suited for investigation of metabolic networks, because it focuses on quantifying individual metabolites without having a bias concerning the choice of targets to be analyzed.
Microbe is one of the best factories in this Earth: efficient, sustainable, and reproducible. Thus, there is considerable intrinsic scientific interest in the question of the regulation of the chemical composition as metabolites. Further, the potential for the application of metabolite engineering in microbial biotechnology is enormous. It is conceivable that through the targeted manipulation of a small number of genes or their regulators it will be possible to improve both yield and quantity for existing and valuable secondary metabolites as potent pharmaceuticals or biomass.
Flavonoids, a highly diverse class of plant secondary metabolites, carry out a broad range of important functions in plants, including roles as pigments, protectants against stresses, and signaling molecules. Recent studies evaluating the health-promoting effects of these plant-derived natural products have generally highlighted their potential in pharmaceutical and nutraceutical applications; however, their limited availability due to seasonal and regional variations and the difficulty inherent to the isolation of single compounds from plants remain intrinsic challenges in this regard. Thus, heterologous gene expression in microbial cell factory is one of the strategy for the industrialized-production of flavonoids. Nucleotide sugars are precursor molecules in biosynthetic pathways of flavonoids. Activated nucleotide sugars are essential donor substrates of glycosyltransferases involved in polyphenol glycosylation processes. Therefore, their availability can influence the glycosylation of flavonoids.
Glycosyltransferases play a critical role in the biosynthesis of therapeutically relevant natural products. Glycosylation significantly affects bioactivity, either through altered interactions with the target binding site, or by altering absorption or pharmacological properties of natural product. The biosynthesis of flavonoids involves specific glycosyltransferases utilizing nucleotide sugars as activated donors. Therefore, precise information about cellular content and metabolic networks of nucleotide sugars is also needed, because the regulation of glycosylation probably includes the availability of nucleotide sugars. Biocatalytic- synthesis is a very attractive way to produce diverse glycosylated polyphenols for medicinal applications. Thus, analysis of nucleotide sugars is essential both in their synthetic production and in studying glycosylation machinery of cells. How the metabolic flux and balance of these diverse nucleotide sugars is regulated, and how this regulation affects the biosynthesis of flavonoids, is far from being understood. Therefore, metabolic and/or pathway engineering of plant-specific plant-origin biosynthesis has attracted an increasing amount of attention.
In this project, we set up in situ quantitative LC-electronspray ionization (ESI)-MS platform, in robust heterologous host Escherichia coli , for a number of nucleotide sugars as the metabolic precursors for glycosylated flavonoids, which are biosynthesized by the expression of plant-origin glycosyltransferase gene together with polyphenol aglycones fed into the host. After the successful visualization of metabolic networks of the nucleotide sugars, we have made the optimized biosynthetic fluxes of nucleotide sugars in the recombinants, yielding the enhanced production of novel bioactive glycosylated flavonoids.
5. We examined several activities with flavonoids and flavonoid glycosides. We found that quercetinq, uercetin-3-glucoside, and '8'-Dihydroxyflavone showed anti-obesity effects in 3T3-L1 cell line-derived adipocyte by decreasing intracellular lipid accumulations and reducing the mRNA expressions involved in adipocyte differentiations. The anti-obesity activities of Quercetin and Quercetin-3-glucoside were also verified in the obese model animals. Based on these results, two patents were applied and two SCI papers are currently under preparation for publication.
Quercetin and Quercetin-3-O-xyloside were shown to activate mouse macrophage cell line, RAW 264.7 cells to secrete TNF-α and IL-6 via activation of NF-kB and MAPKs signaling pathways; Quercetin-3-O-xyloside was shown to be significantly more effective than Quercetin. Currently, a patent and manuscript for SCI publication are under preparation.
Anti-allergic and anti-asthmatic effects and their underlying mechanisms of phloretin, biochanin A, kaempferol, and kaempferol-3-O-rhamnoside on the IgE-antigen complex-mediated allergic responses were evaluated in rat basophilic leukemia RBL-2H3 cells and ovalbumin-induced lung inflammation in asthma model mouse. These compounds were shown to have effective anti-inflammatory and anti-asthmatic activities in both I vitro and in vivo through multiple mechanisms, including a reduction of the levels of Th2 cytokines (IL-4, IL-5 and IL-13), TNF-α via inhibition of Akt and p38 phosphorylation, and IgE. The result of this study was sumitted to an SCI journal and is now under revision. These compounds could be used to develop functional foods and/or therapeutic agents that prevent or/and treat allergic asthma. The results on anti-allergic and anti-asthmatic effects of phloretin and biochanin A were published on Life Sciences (SCI) and those of kaempferol, and kaempferol-3-O-rhamnoside were submitted for publication to another SCI journal.
 표
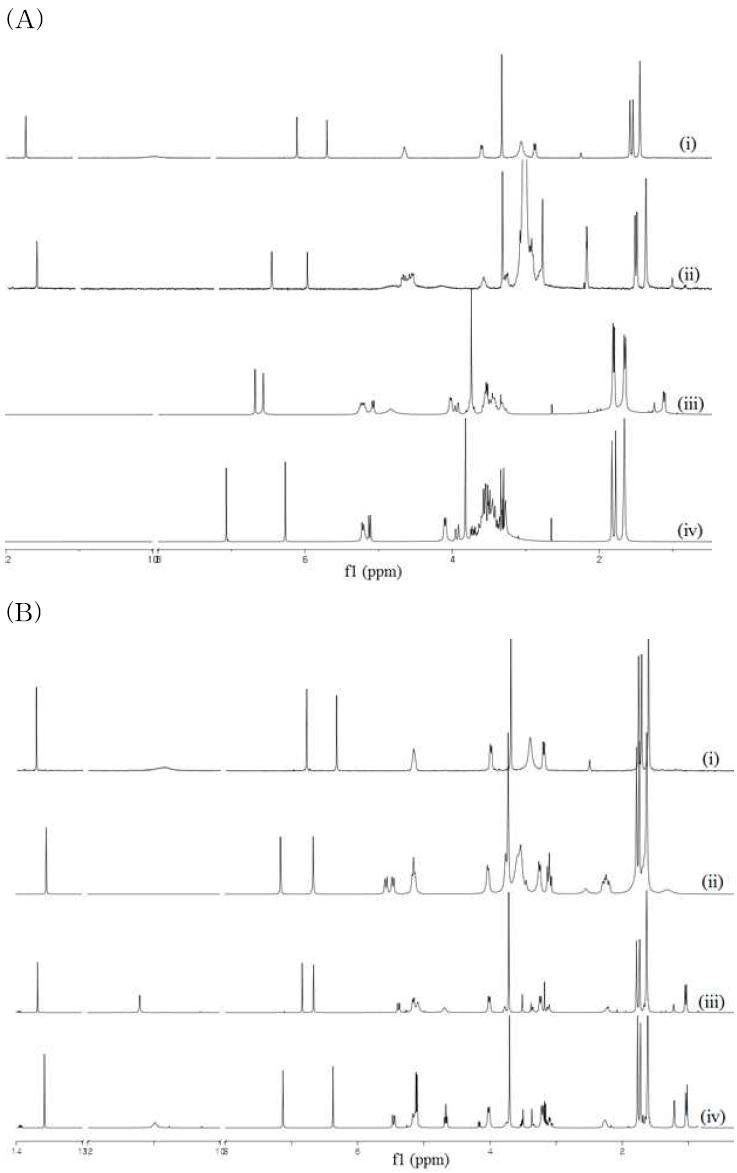
(a) 1HNMRcomparisonofα-mangostin glucopyranosides and α-mangostin standard: (i) a-mangostin; (ii) a-mangostin 3,6-di-O-β-D-glucopyranoside; (iii) a-mangostin 3-O-β-D-glucopyranoside; and (iv) a-mangostin 6-O-β-D- glucopyranoside. (b) 1H NMR comparison of α-mangostin 2-deoxyglucopyranosides and α-mangostin standard: (i) a-mangostin; (ii) a-mangostin 3,6-di-O-β-D-2-deoxy glucopyranoside; (iii) a-mangostin 3-O-β-D-2-deoxyglucopyranoside; and (iv) a-mangostin 6-O-β-D-2-deoxyglucopyranoside.
표
(a) 1HNMRcomparisonofα-mangostin glucopyranosides and α-mangostin standard: (i) a-mangostin; (ii) a-mangostin 3,6-di-O-β-D-glucopyranoside; (iii) a-mangostin 3-O-β-D-glucopyranoside; and (iv) a-mangostin 6-O-β-D- glucopyranoside. (b) 1H NMR comparison of α-mangostin 2-deoxyglucopyranosides and α-mangostin standard: (i) a-mangostin; (ii) a-mangostin 3,6-di-O-β-D-2-deoxy glucopyranoside; (iii) a-mangostin 3-O-β-D-2-deoxyglucopyranoside; and (iv) a-mangostin 6-O-β-D-2-deoxyglucopyranoside.
 표
(a) 1HNMRcomparisonofα-mangostin glucopyranosides and α-mangostin standard: (i) a-mangostin; (ii) a-mangostin 3,6-di-O-β-D-glucopyranoside; (iii) a-mangostin 3-O-β-D-glucopyranoside; and (iv) a-mangostin 6-O-β-D- glucopyranoside. (b) 1H NMR comparison of α-mangostin 2-deoxyglucopyranosides and α-mangostin standard: (i) a-mangostin; (ii) a-mangostin 3,6-di-O-β-D-2-deoxy glucopyranoside; (iii) a-mangostin 3-O-β-D-2-deoxyglucopyranoside; and (iv) a-mangostin 6-O-β-D-2-deoxyglucopyranoside.
표
(a) 1HNMRcomparisonofα-mangostin glucopyranosides and α-mangostin standard: (i) a-mangostin; (ii) a-mangostin 3,6-di-O-β-D-glucopyranoside; (iii) a-mangostin 3-O-β-D-glucopyranoside; and (iv) a-mangostin 6-O-β-D- glucopyranoside. (b) 1H NMR comparison of α-mangostin 2-deoxyglucopyranosides and α-mangostin standard: (i) a-mangostin; (ii) a-mangostin 3,6-di-O-β-D-2-deoxy glucopyranoside; (iii) a-mangostin 3-O-β-D-2-deoxyglucopyranoside; and (iv) a-mangostin 6-O-β-D-2-deoxyglucopyranoside.
| 과제명(ProjectTitle) : | - |
|---|---|
| 연구책임자(Manager) : | - |
| 과제기간(DetailSeriesProject) : | - |
| 총연구비 (DetailSeriesProject) : | - |
| 키워드(keyword) : | - |
| 과제수행기간(LeadAgency) : | - |
| 연구목표(Goal) : | - |
| 연구내용(Abstract) : | - |
| 기대효과(Effect) : | - |
Copyright KISTI. All Rights Reserved.

※ AI-Helper는 부적절한 답변을 할 수 있습니다.