최소 단어 이상 선택하여야 합니다.
최대 10 단어까지만 선택 가능합니다.
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
NTIS 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
DataON 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Edison 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Kafe 바로가기
| 주관연구기관 | 한국지질자원연구원 Korea Institute of Geoscience and Mineral Resources |
|---|---|
| 연구책임자 | 신선명 |
| 참여연구자 | 강정신 , 김철주 , 라제쉬쿠마 , 박진태 , 신동주 , 윤호성 , 이재천 , 이후인 , 정경우 , 최영윤 , 김준수 , 유승준 |
| 보고서유형 | 최종보고서 |
| 발행국가 | 대한민국 |
| 언어 | 한국어 |
| 발행년월 | 2016-12 |
| 과제시작연도 | 2016 |
| 주관부처 | 미래창조과학부 Ministry of Science, ICT and Future Planning |
| 등록번호 | TRKO201700000431 |
| 과제고유번호 | 1711041337 |
| 사업명 | 한국지질자원연구원연구운영비지원 |
| DB 구축일자 | 2017-09-20 |
| 키워드 | 황동광.동광.제련.침출.회수.동.chalcopyrite.copper ore.smelting.leaching.recovery.copper. |
| DOI | https://doi.org/10.23000/TRKO201700000431 |
최종목표
▪ 저품위 황동광 고효율 침출 기술 개발 (침출률 > 85 %)
▪ 저품위 복합동광(산화광/Cu2S)으로부터 산/환원 침출 및동/유가금속 회수 조건 확립
(침출률 > 80%, 동/유가금속 회수율 ≥ 97/80 %)
▪ 건식염화법을 이용한 저품위 동광의 고품위화 공정기술 개발
(황동광 내 철 제거율 ≥ 79 %)
개발내용 및 결과
▪ 제이염화철-염산 및 과산화수소-황산 시스템에서 황동광침출 특성 평가 및 우수 침출률 확보
: 침출률 ≥ 87%(염산+Fe
최종목표
▪ 저품위 황동광 고효율 침출 기술 개발 (침출률 > 85 %)
▪ 저품위 복합동광(산화광/Cu2S)으로부터 산/환원 침출 및동/유가금속 회수 조건 확립
(침출률 > 80%, 동/유가금속 회수율 ≥ 97/80 %)
▪ 건식염화법을 이용한 저품위 동광의 고품위화 공정기술 개발
(황동광 내 철 제거율 ≥ 79 %)
개발내용 및 결과
▪ 제이염화철-염산 및 과산화수소-황산 시스템에서 황동광침출 특성 평가 및 우수 침출률 확보
: 침출률 ≥ 87%(염산+FeCl3)/침출률 ≥ 91%(황산+H2O2)
▪ 황동광 가압 침출 조건 확립
: 침출률 92% 이상 (220℃)
▪ 전해생성 과황산 및 염소 활용 황동광 침출 특성 평가 및 최적 조건 확립
: 침출률 ≥ 85%(과황산)/침출률 ≥ 98%(염소)
▪ 저품위 동광 침출 거동 평가 및 최적 조건 확립
: 침출률 97% 이상 (3M H2SO4, 80℃)
▪ LIX984N을 이용하여 Cu 추출율 99% 이상 달성(15v/v%,향류 3단:모의침출용액, 30v/v%, 향류 3단:고온가압침출용액, 5v/v% 저품위 동광(1.5% Cu)침출용액)
▪ 황화동광의 선택염화반응 메커니즘 규명 및 고품위 chalcocite 제조 : 황화동광의 탈철률 98.0 – 99.8 %
▪ 염화철과 산화동광의 염화반응에 의한 염화구리 및 산화철생성 메커니즘 규명 : 산화동광의 염화율: 92.0 – 99.0 %
기대효과
▪ 저품위 황동광의 습식 제련 기술 확립은 고품위 광석의 고갈에 따른 구리 함유 광석의 저품위화 가속화에 현상에 대한 선제적 대응할 수 방안 확립 및 국내 구리 제련산업의 지속 가능 성장 기반을 마련하는데 기여할 수 있을 것으로 판단됨
적용분야
▪ 저품위 복합광 개발시 기반기술 적용 가능
▪ 황동광의 습식공정 기반기술 확보 및 습득된 단위 기술을 활용한 전략금속 제련기술 개발에 활용 가능
Ⅳ. Results
1. Leaching of copper ore at ambient pressure
◦ Hydrochloric acid leaching
- Hydrochloric acid leaching of chalcopyrite using FeCl3 showed 83% of leaching efficiency of copper at the condition of 0.1M hydrochloric acid, 0.25M FeCl3, leachng temperature o
Ⅳ. Results
1. Leaching of copper ore at ambient pressure
◦ Hydrochloric acid leaching
- Hydrochloric acid leaching of chalcopyrite using FeCl3 showed 83% of leaching efficiency of copper at the condition of 0.1M hydrochloric acid, 0.25M FeCl3, leachng temperature of 90℃, pulp density of 1%(w/v) and leaching time of 6hrs, where leaching reaction is controlled by ash layer diffusion.
- Upon the ultrasonic irradiation, increased leaching efficiency of 87% was obtained, where leaching reaction is controlled by chemical reaction.
◦ Sulfuric acid leaching
- Sulfuric acid leaching of chalcopyrite using H2O2 is much dependent on the stability of H2O2.
- Decomposition of H2O2 in sulfuric acid is markedly suppressed by the addition of ethylene glycol.
- At the condition of 1M H2O2, 2M sulfuric acid, 10mL/L ethylene glycol, leaching temperature of 60℃and leaching time of 240min, 95% of leaching efficiency of copper was obtained.
◦ Pressure leaching of copper ore
- Leaching condition : H2SO4 conc. 5 g/L, oxygen pressure 7 bar, solution 1 liter, pulp density 10%, temperature 230℃, time 1 hour, agitation speed 500rpm
- Cu leaching efficiency 100%, iron conc. in leaching solution 0.18g/L, pH of leaching solution 0.70
- Behaviour of residue
· main minerals : hematite와 SiO2 / particle size : - 30㎛ hematite, + 30㎛ SiO2
· filtration of residue : very good
◦ Leaching of copper ore by use of electrogenerated Cl2
- Upon leaching of chalcopyrite in NaCl(0.5 ~ 2M) and Sulfuric acid(0.5 ~ 6M) solutions at pulp density of 15(w/v) and temperature of 25℃ by use of electrogenerated Cl2 as an oxidant, very high leaching efficiency of 98% for copper is obtained
- Leaching yield of copper is much affected by the supply rate of Cl2, however, is not dependent on the concentration of sodium chloride or sulfuric acid and the temperate of leaching solution.
◦ Leaching of copper ore by use of electrogenerated S2O82-
- Upon leaching of chalcopyrite in sulfuric acid solution by use of S2O82- as an oxidant, leaching yield of shows the value of more than 50%. The addition of Clions in to leaching solution increases the leaching efficiency, which is due to the suppression in passivation by sulfur of by-product and higher leaching efficiency than 90% for copper is obtained upon leaching of chalcopyrite in hydrochloric acid solution by use of S2O82-
- S2O82- is effectively generated in the solution of 0.3M Na2SO4 and 2.7M (NH4)2SO4 by the apply of current(500mA/cm2) and leaching efficiencies of 70% and 85% for copper are observed in the absence and presence of Cl- ions, respectively upon leaching of chalcopyrite in sulfuric acid solution by use of electrogenerated S2O82-
2. Separation and extraction of copper from chalcopyrite leachant
◦ Cu, Fe Separately cementation from copper ore simulated leachant
- Fe2+and Cu2+ are co-precipitated in pH 2.5∼4.0 and Fe3+ is previously precipitated in pH 2.0∼2.5
- When C2H2O4 is put into the leachant, Cu is precipitated as CuC2O4 (Efficiency of precipitation: Cu 99%, Fe 5%)
- When NaBH4 is put into the leachant, Cu is precipitated as Cu and Cu2O.
◦ Solvent Extraction of Cu from copper ore simulated leachant
- By using 20v/v% LIX 984N as extractant, initial feed pH 1.5, O/A ratio 2, 2 stage counter current extraction: Cu 99.8%, Fe 0.6% as extraction efficiency 3 stage counter current extraction: Cu 99.3%, Fe 0.6% as extraction efficiency
◦ Separation of Cu, Fe from high temperature – pressure chalcopyrite ore leachant
- When NaBH4 and C2H2O4 are put into the leachant, Cu is precipitated above 99%.
- By using 30v/v% LIX 984N as extractant, Cu is extracted above 99.3% and other metals are extracted below 0.2%.
3. Leaching and separation/extraction of copper from complex copper ore
◦ Leaching of complex copper ore
- Preparation of 1.5% Cu sample for leaching test by liberation.
- Leaching tests with H2SO4 concentration and temperature(optimum condition: 3M H2SO4, 80℃, Leaching efficiency of Cu: 97%)
◦ Separation and extraction of Cu from complex copper ore
- Solvent extraction test with LIX 984N concentration (Initial feed pH 1.5, O/A ratio
1): Extraction efficiency of Cu 98%
- Above 20 v/v% LIX 984N, Fe is co-extracted about 2∼3%. Therefore multi stage count-current solvent extraction is carried out below 15 v/v% LIX 984N.
- 2 stage count-current solvent extraction by 10, 15 v/v% LIX 984N has problem(emulsion occurrence)
- 2 stage count-current solvent extraction by 5 v/v% LIX 984N: Extraction efficiency of Cu 98%, Fe 1.7ppm
4. Production of high-grade chalcocite from low-grade Cu-sulfide ore
◦ Thermodynamic analysis using chemical potential diagrams of Cu-S-Cl and Fe-S-Cl systems at 1100 K was conducted. The results showed that high-grade chalcocite can be obtained through the selective removal of iron from low-grade Cu-sulfide ore when cuprous chloride is used as a chlorinating agent. In addition, the feasibility of the selective chlorination process was demonstrated by the experiments.
◦ When Cu-sulfide ore was reacted with cuprous chloride at 800 – 1100 K for 5 h in the presence or absence of sulfur under an Ar gas atmosphere, the concentration of iron in Cu-sulfide ore was decreased from 23.6 – 31.5 % to below 0.37 % and the concentration of copper was increased from 34.6 – 39.8 % to 58.7 – 78.3 %, and high-grade chalcocite was produced.
◦ Iron removal ratio by the selective chlorination process using CuCl for Cu-sulfide ore was 98.0 – 99.8 %
◦ Thermodynamic analysis using chemical potential diagrams of Cu-O-Cl and Fe-O-Cl systems at 1100 K and vapor pressure diagram of some selected chlorides and oxides at the elevated temperatures was conducted. The results showed that iron oxide and cuprous chloride can be obtained through the chlorine recovery process by reacting ferrous chloride and Cu-oxide ore. In addition, the feasibility of the chlorine recovery process was demonstrated by the experiments.
◦ When Cu-oxide ore was reacted with ferrous chloride at 1000 – 1100 K for 1 – 9 h under an Ar gas atmosphere, the concentration of copper in Cu-oxide ore was decreased from 6.03 – 7.70 % to 0.06 – 0.53 % and the concentration of iron was increased from 1.55 – 2.20 % to 5.10 – 35.9 %, and cuprous chloride and iron oxide were obtained after the chlorine recovery process
◦ Chlorination ratio of the Cu-oxide ore by the chlorine recovery process using ferrous chloride was 92.0 – 99.0 %
 표
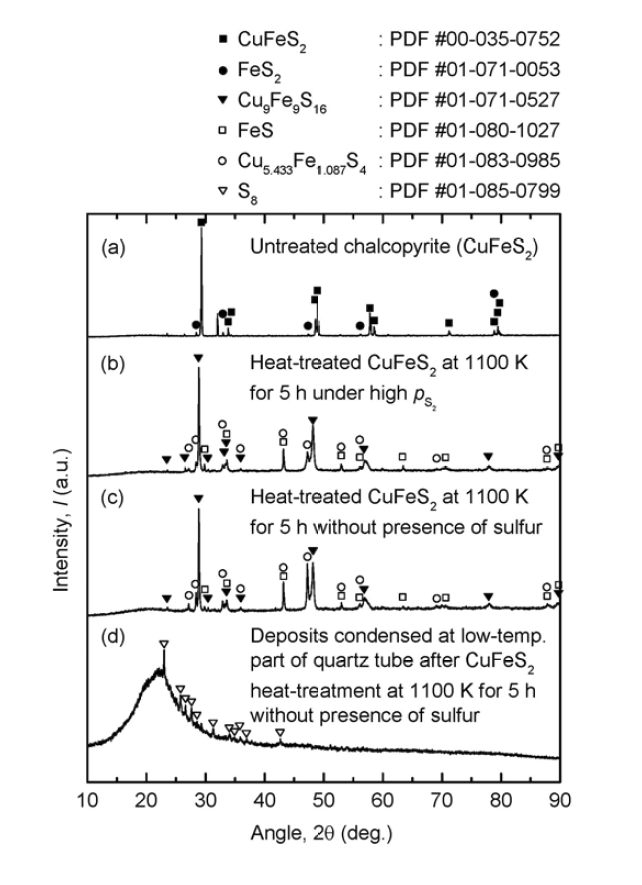
Results of XRD analysis for (a) untreated chalcopyrite and heat-treated chalcopyrite at 1100 K for 5 h (b) in the presence of sulfur, (c) in the absence of sulfur, and (d) deposits condensed at the low-temperature part of the quartz tube after the decomposition of chalcopyrite at 1100 K for 5 h in the absence of sulfur.
표
Results of XRD analysis for (a) untreated chalcopyrite and heat-treated chalcopyrite at 1100 K for 5 h (b) in the presence of sulfur, (c) in the absence of sulfur, and (d) deposits condensed at the low-temperature part of the quartz tube after the decomposition of chalcopyrite at 1100 K for 5 h in the absence of sulfur.
 표
Results of XRD analysis for (a) untreated chalcopyrite and heat-treated chalcopyrite at 1100 K for 5 h (b) in the presence of sulfur, (c) in the absence of sulfur, and (d) deposits condensed at the low-temperature part of the quartz tube after the decomposition of chalcopyrite at 1100 K for 5 h in the absence of sulfur.
표
Results of XRD analysis for (a) untreated chalcopyrite and heat-treated chalcopyrite at 1100 K for 5 h (b) in the presence of sulfur, (c) in the absence of sulfur, and (d) deposits condensed at the low-temperature part of the quartz tube after the decomposition of chalcopyrite at 1100 K for 5 h in the absence of sulfur.
| 과제명(ProjectTitle) : | - |
|---|---|
| 연구책임자(Manager) : | - |
| 과제기간(DetailSeriesProject) : | - |
| 총연구비 (DetailSeriesProject) : | - |
| 키워드(keyword) : | - |
| 과제수행기간(LeadAgency) : | - |
| 연구목표(Goal) : | - |
| 연구내용(Abstract) : | - |
| 기대효과(Effect) : | - |
Copyright KISTI. All Rights Reserved.

※ AI-Helper는 부적절한 답변을 할 수 있습니다.