최소 단어 이상 선택하여야 합니다.
최대 10 단어까지만 선택 가능합니다.
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
NTIS 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
DataON 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Edison 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Kafe 바로가기
| 주관연구기관 | 전북대학교 Chonbuk National University |
|---|---|
| 연구책임자 | 장점석 |
| 참여연구자 | 주진 , 조민 , 카밀라칸난 , 강은정 , 서성아 , 오현희 , 이경림 , ANNAMALAIALAGAPPAN , LOGANATHAN PRABURAMAN , 임정묵 , 조항욱 , MAHADIK MAHADEO ABASAHEB , SHINDE PRAVIN SHRIPATI , 김수경 , 이경근 , 서영석 , 이정열 , 안길우 , 황준하 , SELVARAJ DAVID , 박진우 |
| 보고서유형 | 최종보고서 |
| 발행국가 | 대한민국 |
| 언어 | 한국어 |
| 발행년월 | 2017-05 |
| 과제시작연도 | 2016 |
| 주관부처 | 환경부 Ministry of Environment |
| 등록번호 | TRKO201700018019 |
| 과제고유번호 | 1485013892 |
| 사업명 | 환경정책기반공공기술개발사업 |
| DB 구축일자 | 2018-03-03 |
| 키워드 | 황화수소.태양광.수소제조.광전극.광전기화학 시스템.H2S.Solar light.H2 production.Photoelectrode.PEC system. |
| DOI | https://doi.org/10.23000/TRKO201700018019 |
개발 목적 및 필요성
본 연구는 태양광에너지 전환물질인 이종접합형 나노구조체 광전극 소재를 이용하여 석유정제 공정 내 폐가스(황화수소)처리와 동시에 청정에너지 회수를 위한 신공정 개발을 목적으로(태양에너지 전환효율 4%이상 달성) 한다. 기존에 개발된 광촉매 및 광전극 소재들은 주로 입자형태이거나 박막형 형태가 대부분을 차지한다. 그러므로, 입자간의 저항을 줄이고, 빛의 이용율을 향상시키기 위해 특정 기질 위에 한방향으로 직접 성장된 광전극 소재를 개발하고자 한다.
연구개발결과
1. 1차원 나노구조 광전극의
개발 목적 및 필요성
본 연구는 태양광에너지 전환물질인 이종접합형 나노구조체 광전극 소재를 이용하여 석유정제 공정 내 폐가스(황화수소)처리와 동시에 청정에너지 회수를 위한 신공정 개발을 목적으로(태양에너지 전환효율 4%이상 달성) 한다. 기존에 개발된 광촉매 및 광전극 소재들은 주로 입자형태이거나 박막형 형태가 대부분을 차지한다. 그러므로, 입자간의 저항을 줄이고, 빛의 이용율을 향상시키기 위해 특정 기질 위에 한방향으로 직접 성장된 광전극 소재를 개발하고자 한다.
연구개발결과
1. 1차원 나노구조 광전극의 합성
불소가 첨가된 산화 주석(FTO)을 코팅한 유리 위에 성장한 후 500°C에서 열처리한 TiO2나노막대와 FTO 유리 위의 Fe2O3 나노막대는 간단한 수열합성을 사용하여 성장시켰다.
2. 헤테로접합 광전극 물질들의 합성.
1) 본 연구그룹은 ZIS 전구체 농도를 조절하고 2단계 수열합성 방법을 사용하여 1차원 TiO2 NR 위에 계면활성제가 없는 푹신한 2차원 ZIS NS을 성공적으로 합성하였다.
2) 간단한 2단계 수열합성 공정과 에틸렌디아민 (EDA)같은 용액과 주형 그리고 조정제를 응용하여 새로운 광전극 구조인 카드뮴 설파이드(CdS) 나노플라워/루타일(R)-TiO2 나노막대(NR) 헤테로접합을 성공적으로 합성하였다.
3) 새로운 3차원 구조의 CdS/ZnIn2S4/TiO2 헤테로접합은 3단계의 간단한 수열 합성 방법으로 만들어졌다. 삼차원 헤테로접합을 형성하기 위해서 TiO2 나노막대 위에 ZnIn2S4을 설계하고 CdS 나노 입자로 감광시켰다.
4) 수열합성 방법을 통한 FTO위에 향상된 광전기화학적 CdS/Zr:α-Fe2O3 나노구조체 배열을 직접적으로 합성했다.
3. 보조 촉매의 개발/헤테로접합 광전극 물질
간단하고 효과적인 전략은 헤테로구조의 Ni(OH)2/CdS/ZnIn2S4/TiO2 광전극의 성능을 근본적으로 향상시키기 위해 사용되었다.
4. Gradient Core/Shell Quantum Dot Photocatalyst의 합성
CdS 기반의 물질 연구들 중에 4번 코팅된 CdS/ZnS는 CdS와 3번 코팅된 CdS/ZnS에 비해 눈에 띄게 전체적으로 향상된 광전류를 나타내었다.
5. 대면적 광전기화학 전극으로 구성된 시스템 개발
(1) 대면적 광전극 제작 (10x10cm2):
(a) CdS 나노플라워/TiO2 나노로드
(b) Al2O3/CdS/Zr:Fe2O3 나노로드
(2) 옥내외에서 평가가 가능한 광전기화학적 시스템 설계 및 제작
광전극 셀을 설치한 광전기화학 시스템을 구축하였다. 10x10cm2으로 제작된 광전극 (working electrode)와 상대전극(Pt 전극)을 광전극 셀에 고정화하였으며, 이를 활용하여 광전기화학 시스템과 연결하였다.
성능사양 및 기술개발 수준
본 연구에서는 다양한 광전극 소재들이 개발되었으며, 가장 우수한 성능을 보이는 두 물질로는 태양광전환(STH(%)) 효율이 3.95%인 CdS NF/R-TiO2 NR 소재와 효율이 6.22%인 Al2O3/CdS/Zr:Fe2O3 광전극 소재를 확보할 수 있었다.
활용계획
- H2S 광분해용 광전극 소재 개발에 대한 유용한 기술적 정보 제공
- 유해성 환경 오염물질분야(수질정화, 살균/항균, 공기정화, 악취제거)에 활용
- 태양전지의 전극소재 개발에 대한 합성기술 제공
(출처 : 요약서 3p)
Ⅳ. Results
1. Synthesis of one-dimensional nanostructure photoanode
1) TiO2 Nanorod/FTO Substrate
TiO2 nanorods grown hydrothermally on fluorine-doped tin oxide (FTO) and annealed at 500 °C yielded an optimal photocurrent density of ~988 μA cm-2 at 0.1 V vs.
Ⅳ. Results
1. Synthesis of one-dimensional nanostructure photoanode
1) TiO2 Nanorod/FTO Substrate
TiO2 nanorods grown hydrothermally on fluorine-doped tin oxide (FTO) and annealed at 500 °C yielded an optimal photocurrent density of ~988 μA cm-2 at 0.1 V vs. Ag/AgCl under simulated illumination conditions. The annealing process minimized the defects in TiO2 and assisted in the formation of close contacts between the FTO and TiO2 nanorods, which allowed for efficient electron transport.
2) Zr:Fe2O3 Nanorod/FTO Substrate
Hematite nanorods on FTO glass were deposited on FTO substrate by using a simple hydrothermal method as previously reported. the photocurrent density of the pristine 1D Zr:α-Fe2O3 is ca. 0.75 mA cm-2 at -0.3 V Ag/AgCl. In the next work, Metal oxides formed a perfectly uniform shell layer on the outside of the 1D Zr:Fe2O3. That is, the 1D Zr:Fe2O3 nanorod will play a role of buliding block structure as well as visible active materials.
2. Synthesis of heterojunction photoanode materials
1) ZnIn2S4 Nanosheet/TiO2 Nanorod
We have successfully prepared the surfactant free fluffy 2D ZIS NS’s onto the 1D TNR’s using conventional two step hydrothermal method by controlling the concentration of ZIS precursor. The empirical measurements were used to confirm that the manifold concentration of ZIS precursor is one of the crucial factor to determining the morphologies, wavelength, and crystal phase. The PEC performance for the entire samples attained manifold enhancement due to the effect of concentration of the ZIS precursor. The optimized heterojunction photoanode reveal higher photocurrent density is 1600 μA/Cm2 at an applied potential of –0.5V vs .Ag/AgCl under the BSI, which is 3 fold higher than the bare 1D TNR’s.
2) CdS Nanoflower/TiO2 Nanorod
With the application of ethylenediamine (EDA) as a solvent, a template, and co-ordination agents, novel photoanode architecture of the cadmium sulfide (CdS) nanoflower (NF)/rutile(R)-TiO2 nanorod(NR) heterojunction is successfully synthesized using a facile two-step hydrothermal process.The optimized CdS(medium concentration; MC)NF/R-TiO2 NR heterojunction exhibited agreatly enhanced visible-light photoelectrochemical (PEC) performance, whereby the highest photocurrent density of 3.23 mA cm-2 at 0.1V versus Ag/AgCl and a photoconversion efficiency(PCE) of 0.46% were achieved under solar light irradiation. The optimal photocurrent density of the CdS (MC) NF/R-TiO2 NR heterojunction, photoanode is 2.54 times(60.68%) higher than that of the pristine R-TiO2 NR. this surface modification reveals the synergistic effect. The optimal CdS(MC)NF/R-TiO2 NR heterojunction showed a PEC hydrogen (H2) generation of 1007.98 μmol after 3hr. The design of the novel heterojunction photoanode that is proposed in the present strategy canshed light on the fabrication of new,cheap photocatalysts for an effective H2generation.
3) CdS/ZnIn2S4/TiO2 Heterojunction
A novel, three-dimensional, ternary CdS/ZnIn2S4/TiO2 heterojunction has been fabricated via a three-step facile hydrothermal method. In this structure, one-dimensional TiO2 nanorods were directly grown on conductive fluorine-doped tin oxide (FTO) substrates. Next, to form a ternary heterojunction of CdS/ZnIn2S4/TiO2, ZnIn2S4 nanosheets were designed on the TiO2 nanorods and sensitized by CdS nanograins. A systematic photoelectrochemical study shows that the photocurrent density of the ternary heterojunction architecture is as high as 1.4 mA cm-2 at a potential of 0.1 V versus Ag/AgCl (under optimized conditions). A more detailed study shows that the photocurrent density is more than two times higher than that of a single CdS/TiO2 heterojunction (0.615 mA cm-2) and three times higher than that of ZnIn2S4/TiO2 (at 0.1 V vs. Ag/AgCl). This excellent photoelectrochemical performance is ascribed to the way that the band structure of TiO2 nanorods synergistically cascades with ZnIn2S4 and CdS, which allows for the absorption of a wider portion of the solar spectrum and improves the effective separation of the generated electron–hole pairs. Electrochemical impedance spectroscopy (EIS) studies also reveal the significant changes in both the interface resistance and the charge transfer resistance of the CdS/ZnIn2S4/TiO2 heterostructure. This can be attributed to the efficient hierarchical cascading that occurs during the electron–hole transfer from the excited CdS to TiO2 through the enlarged interface of ZnIn2S4 upon visible light illumination.
4) CdS/Zr:Fe2O3 Nanorod
Well-defined CdS nanograin-sensitized one-dimensional (1D) Zr:α-Fe2O3 nanostructured arrays with enhanced photoelectrochemical performance are synthesized directly on F-doped SnO2 (FTO) using the hydrothermal method. Owing predominantly to the appropriate photogenerated electron–hole separation and charge collection in 1D Zr:α-Fe2O3 nanorods, hydrothermally deposited CdS/1D Zr:α-Fe2O3 samples exhibit improved photocurrent density over CdS/Fe2O3 nanosheets prepared by other methods. In our work, compared with 1D Zr:α-Fe2O3, the CdS-sensitized 1D Zr:α-Fe2O3 nanorod arrays show 1.9 times improved photoelectrochemical performance. Unfortunately, CdS nanograin-sensitized 1D Zr:α-Fe2O3 nanorod arrays suffer from instability problem. Nickel hydroxide loading, however, can boost the photoelectrochemical performance of the heterojunction and also act as a protective layer that improves the stability of the Ni(OH)2/CdS/1D Zr:α-Fe2O3 electrode compared to CdS/1D Zr:α-Fe2O3.
3. Development of Cocatalyst/Heterojunction photoanode materials
1) Ni(OH)2/CdS/ZnIn2S4/TiO2 Heterojunction
A simple and effective strategy was used to fundamentally improve the performance of a heterostructured Ni(OH)2/CdS/ZnIn2S4/TiO2 photoanode. TiO2 nanorods grown hydrothermally on fluorine-doped tin oxide (FTO) and annealed at 500℃ yielded an optimal photocurrent density of ~988 μA cm-2 at 0.1 V vs. Ag/AgCl under simulated illumination conditions. The annealing process minimized the defects in TiO2 and assisted in the formation of close contacts between the FTO and TiO2 nanorods, which allowed for efficient electron transport. The stepwise introduction of ZnIn2S4 and CdS on annealed TiO2 enhanced absorption in the visible range and electron/hole separation in CdS/ZnIn2S4/TiO2. Additionally, the Ni(OH)2 cocatalyst functioned in hole trapping and improved the stability of the photoelectrode through timely consumption of the photogenerated charges, particularly the holes.
4. Synthesis of Gradient Core/Shell Quantum Dot Photocatalyst
1) CdZnS/ZnS Core/Shell Quantum Dot
Among the studied CdS based samples, the CdS/ ZnS 4times demonstrates remarkably enhanced photocurrent compared to the CdS and CdS/CdS/ZnS 3times over the entire potential window. Each J-V graph shows four light curves under light condition taken at four separate scan. It was ssen that after first scan measurement photocurrent decrses and then for the next three cycles it is nearly same. The presence of ZnS leads to the enhancement of transient photocurrent response of CdS from 384 μA cm-2 to 698 μAcm-2 for CdS/ZnS 4 times), almost 2 times higher than that of the pure CdS. Moreover, the transient photocurrents were rapid, steady and reproducible during three switched on–off cycles. When the light is interrupted during chopping condition, the accumulated holes in the surface state gradually vanish, and a spike is observed. The initial decay of the photocurrent indicates that a fraction of h+ which reaches the semiconductor surface, instead of capturing e−from the electrolyte, either accumulates at the surface or recombines with electrons from the conduction band. After recombination of the excessive h+, the generation and transfer of electron−hole pairs reach equilibration ,and a constant current is formed. Upon light illumination, the photocurrent increases sharply and the current intensity returns quickly to the dark current state when the light is turned off. With the addition of ZnS on CdS, the heterostructure photocatalysts have lower recombination of electron–hole pairs than pure CdS, which promotes the more effective migration of charge-carriers and the photocatalytic activity.
5. System Construction consisting of Large Sized Heterojunction Photoanode
(1) Fabrcation of Large Sized Photoanode (10x10cm2):
(a) CdS Nanoflower/TiO2 Nanorod
(b) Al2O3/CdS/Zr:Fe2O3 Nanorod
(2) System construction consisting of large sized photoanode for inoor and outdoor test
(출처 : Summary 18p)
 표
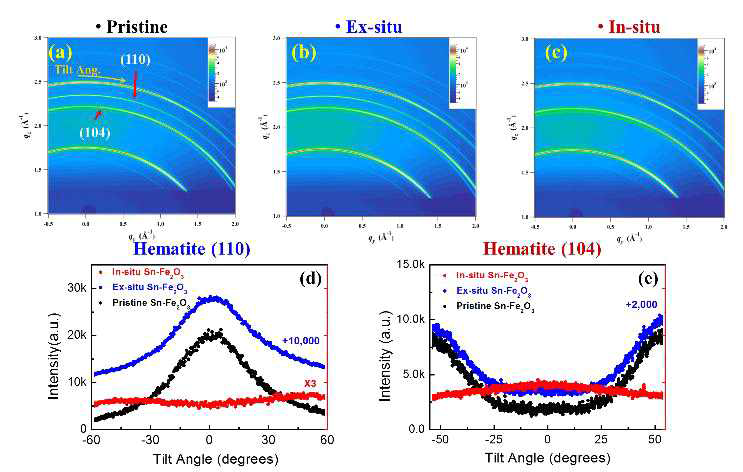
(a) –(c) are 2D XRD images of the pristine(black dot), Ex-situ (blue dot), and In-situ (red dot) Sn-doped hematite photoanodes annealed at 800˚C, respectively. (d)-(e) The circular line cuts passing the hematite (110) and (104) peaks in which zero degree corresponds to the surface normal direction.
표
(a) –(c) are 2D XRD images of the pristine(black dot), Ex-situ (blue dot), and In-situ (red dot) Sn-doped hematite photoanodes annealed at 800˚C, respectively. (d)-(e) The circular line cuts passing the hematite (110) and (104) peaks in which zero degree corresponds to the surface normal direction.
 표
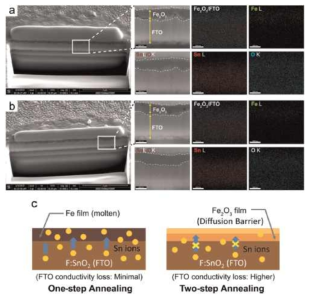
Cross-sectional scanning electron microscopic images of α-Fe2O3 photoanodes prepared by (a)one-step and (b) two-step annealing method.The textured regionis represented by a dark-field scanning electron microscopy images at a magnification of 150k and corresponds to the elemental maps of Fe, Sn, and O. The samples were partly melted due to the high energy of FIB during the sample preparation. (c) Schematic illustration of growth mechanism during the one-and two-step annealing processes.
표
Cross-sectional scanning electron microscopic images of α-Fe2O3 photoanodes prepared by (a)one-step and (b) two-step annealing method.The textured regionis represented by a dark-field scanning electron microscopy images at a magnification of 150k and corresponds to the elemental maps of Fe, Sn, and O. The samples were partly melted due to the high energy of FIB during the sample preparation. (c) Schematic illustration of growth mechanism during the one-and two-step annealing processes.
 표
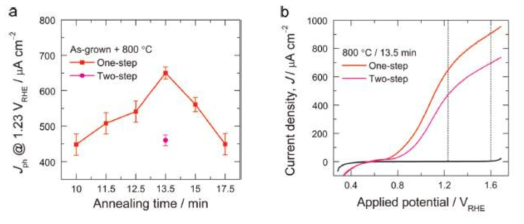
(a) Jph variation of one-step annealed hematite photoanodes measured at 1.23 VRHE in 1M NaOH for different annealing times. For comparison, Jph value at 1.23V RHE of two-step-annealed α-Fe2O3 photoanode prepared by similar higher temperature annealing condition is shown. (b) J–V curves of one- and two-step annealed hematite photoanodes with similar high temperature annealing at 800 ˚C for 13.5 min. The error bars represent the standard deviations in PEC measurements of the independently prepared series of hematite photoanodes. Light illumination, 1sun; scan rate, 50mVs-1.
표
(a) Jph variation of one-step annealed hematite photoanodes measured at 1.23 VRHE in 1M NaOH for different annealing times. For comparison, Jph value at 1.23V RHE of two-step-annealed α-Fe2O3 photoanode prepared by similar higher temperature annealing condition is shown. (b) J–V curves of one- and two-step annealed hematite photoanodes with similar high temperature annealing at 800 ˚C for 13.5 min. The error bars represent the standard deviations in PEC measurements of the independently prepared series of hematite photoanodes. Light illumination, 1sun; scan rate, 50mVs-1.
 표
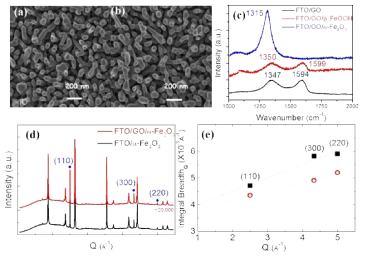
FE-SEM images of photoanodes (a) without and (b) with a GO underlayer.(c) Raman spectra of FTO/GO, FTO/GO/β-FeOOH, and FTO/GO/α-Fe2O3 photoanodes sintered at 800˚C. (d) Synchrotron X-ray diffraction patterns of α -Fe2O3(marked in blue) hotoanodes on FTO substrates(remaining peaks) sintered at 800oC without (black line) and with a GO(red line) interlayer.(e) Williamson-Hall plots of the hematite peaks denoted as blue dots in (d). The dashed lines represent linear regressions.
표
FE-SEM images of photoanodes (a) without and (b) with a GO underlayer.(c) Raman spectra of FTO/GO, FTO/GO/β-FeOOH, and FTO/GO/α-Fe2O3 photoanodes sintered at 800˚C. (d) Synchrotron X-ray diffraction patterns of α -Fe2O3(marked in blue) hotoanodes on FTO substrates(remaining peaks) sintered at 800oC without (black line) and with a GO(red line) interlayer.(e) Williamson-Hall plots of the hematite peaks denoted as blue dots in (d). The dashed lines represent linear regressions.
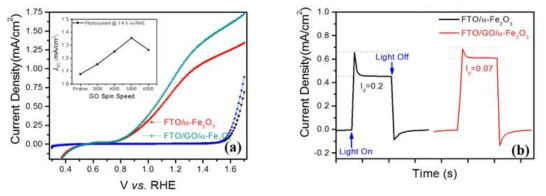 표
(a) Photocurrent-potential (J-V) curves and (b) Transient photocurrent measurement for the PEC water oxidation reaction with and without GO-based α-Fe2O3 photoanodes in FTO substrates sintered at 800˚C. 1M NaOH was used under 1sun standard illumination conditions and the inset shows the photocurrent densities of the hematite photoanodes with and without GO underlayers at 1.4V vs. RHE under 1 sun standard illumination conditions.
표
(a) Photocurrent-potential (J-V) curves and (b) Transient photocurrent measurement for the PEC water oxidation reaction with and without GO-based α-Fe2O3 photoanodes in FTO substrates sintered at 800˚C. 1M NaOH was used under 1sun standard illumination conditions and the inset shows the photocurrent densities of the hematite photoanodes with and without GO underlayers at 1.4V vs. RHE under 1 sun standard illumination conditions.
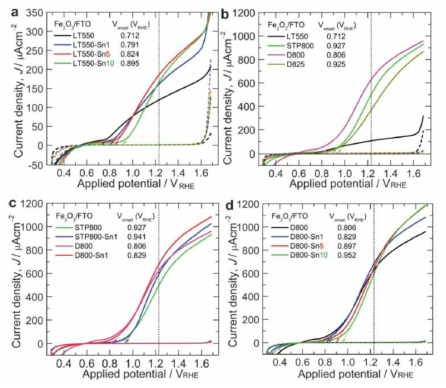 표
Current density–voltage (J–V) curves of (a) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by LT-annealing at 550 1C, (b) pristine a-Fe2O3 photoanodes fabricated under different annealing conditions, (c) pristine and 1 mM Sn-doped a-Fe2O3 photoanodes fabricated at high temperature by two-step and one-step annealing methods, and (d) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by one-step HT-annealing at 800 1C. The tangent arrows pointing toward the X-axis gives the onset potential (Vonset) of all the a-Fe2O3 photoanodes.
표
Current density–voltage (J–V) curves of (a) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by LT-annealing at 550 1C, (b) pristine a-Fe2O3 photoanodes fabricated under different annealing conditions, (c) pristine and 1 mM Sn-doped a-Fe2O3 photoanodes fabricated at high temperature by two-step and one-step annealing methods, and (d) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by one-step HT-annealing at 800 1C. The tangent arrows pointing toward the X-axis gives the onset potential (Vonset) of all the a-Fe2O3 photoanodes.
 표
Nyquist plots (real vs. imaginary impedance) of (a) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by LT-annealing at 550 1C, (b) pristine a-Fe2O3 photoanodes fabricated under different annealing conditions, (c) pristine and 1 mM Sn-doped a-Fe2O3 photoanodes fabricated at a high temperature by two-step and one-step annealing methods, and (d) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by one-step HT-annealing at 800 1C. The EIS data were obtained at a bias potential of 1.23 VRHE under 1 sun illumination in 1 M NaOH. The discrete symbols and the solid lines/curves in the plots represent the experimental data and the results of fitting, respectively. The inset of (a) shows the equivalent electrochemical circuit used for fitting all the Nyquist plots.
표
Nyquist plots (real vs. imaginary impedance) of (a) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by LT-annealing at 550 1C, (b) pristine a-Fe2O3 photoanodes fabricated under different annealing conditions, (c) pristine and 1 mM Sn-doped a-Fe2O3 photoanodes fabricated at a high temperature by two-step and one-step annealing methods, and (d) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by one-step HT-annealing at 800 1C. The EIS data were obtained at a bias potential of 1.23 VRHE under 1 sun illumination in 1 M NaOH. The discrete symbols and the solid lines/curves in the plots represent the experimental data and the results of fitting, respectively. The inset of (a) shows the equivalent electrochemical circuit used for fitting all the Nyquist plots.
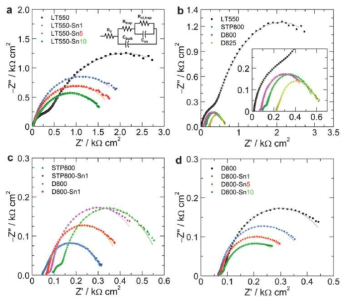
 표
(a) Jph variation at 1.23 VRHE for PRED-treated Fe2O3/Fe photoanodes prepared at 800˚C with different annealing times. The error bars represent the standard deviations in J–V measurements. The inset of (a) shows the J–V curves of bare and PRED-treated Fe2O3/Fe photoanodes annealed at 800˚C for 6 min, and (b) shows the corresponding photo-stability curves. Magnified portions show the current-responses under chopped illumination (100 mW cm−2) at 1.23 VRHE in 1 M NaOH.
표
(a) Jph variation at 1.23 VRHE for PRED-treated Fe2O3/Fe photoanodes prepared at 800˚C with different annealing times. The error bars represent the standard deviations in J–V measurements. The inset of (a) shows the J–V curves of bare and PRED-treated Fe2O3/Fe photoanodes annealed at 800˚C for 6 min, and (b) shows the corresponding photo-stability curves. Magnified portions show the current-responses under chopped illumination (100 mW cm−2) at 1.23 VRHE in 1 M NaOH.
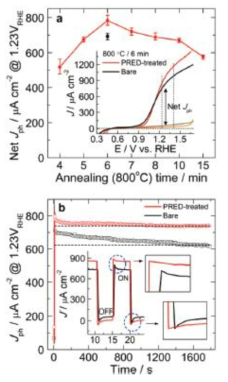
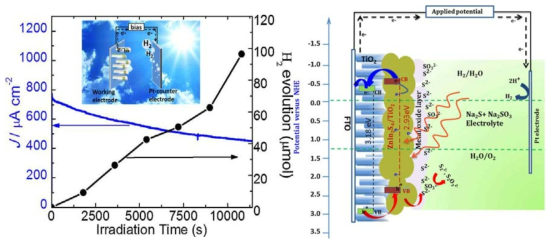 표
A. Schematic of charge transfer mechanism in metal oxide coated ZT photoanode. B. 28. Evolution of H2 and photocurrent density as a function of time measured at 0.1 V vs. Ag/AgCl using SZT as a photoelectrode under 100mW cm−2 light illumination in a three-electrode configuration, Inset shows a schematic of the photoelectrochemical hydrogen generation.
표
A. Schematic of charge transfer mechanism in metal oxide coated ZT photoanode. B. 28. Evolution of H2 and photocurrent density as a function of time measured at 0.1 V vs. Ag/AgCl using SZT as a photoelectrode under 100mW cm−2 light illumination in a three-electrode configuration, Inset shows a schematic of the photoelectrochemical hydrogen generation.
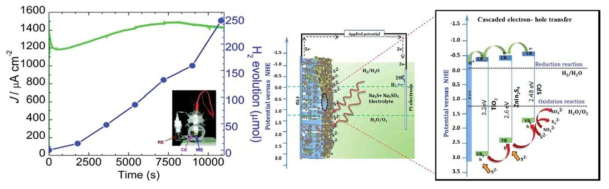 표
A. Schematic mechanism of the cascading electron and hole transfer in the ternar CdS/ZnIn2S4/TiO2 heterojunction system. B. Evolution of H2 and photocurrent density as a function of time form the CdS/ZnIn2S4/TiO2 photoanode under 100 mW cm2 light illumination. The inset shows a picture of the simple photoelectrochemical cell setup used for hydrogen generation (WE: working electrode, CE: counter electrode, and RE: reference electrode).
표
A. Schematic mechanism of the cascading electron and hole transfer in the ternar CdS/ZnIn2S4/TiO2 heterojunction system. B. Evolution of H2 and photocurrent density as a function of time form the CdS/ZnIn2S4/TiO2 photoanode under 100 mW cm2 light illumination. The inset shows a picture of the simple photoelectrochemical cell setup used for hydrogen generation (WE: working electrode, CE: counter electrode, and RE: reference electrode).
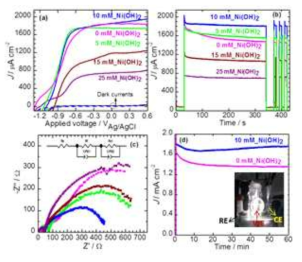 표
(a) Photocurrent density – potential characteristics, (b) long-term photostability and transient photocurrent responses, (c) EIS plots of CdS/ZnIn2S4/TiO2 photoelectrodes prepared with different nickel precursor concentrations(5, 10, 15 and 25 mM). Inset shows the equivalent circuit, and (d) Photocurrent stability measurement of a CdS/ZnIn2S4/TiO2 and 10mM Ni(OH)2/CdS/ZnIn2S4/TiO2 heterojunctionphotoelectrodesat an applied potential of 0.1 V (vs Ag/AgCl) for 1 h.
표
(a) Photocurrent density – potential characteristics, (b) long-term photostability and transient photocurrent responses, (c) EIS plots of CdS/ZnIn2S4/TiO2 photoelectrodes prepared with different nickel precursor concentrations(5, 10, 15 and 25 mM). Inset shows the equivalent circuit, and (d) Photocurrent stability measurement of a CdS/ZnIn2S4/TiO2 and 10mM Ni(OH)2/CdS/ZnIn2S4/TiO2 heterojunctionphotoelectrodesat an applied potential of 0.1 V (vs Ag/AgCl) for 1 h.
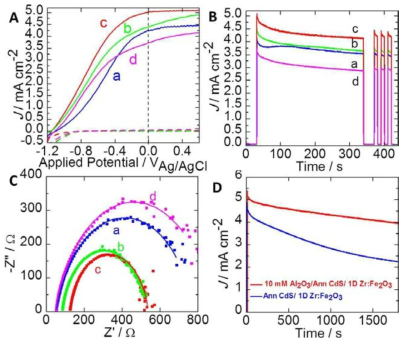 표
A) Linear sweep voltammetric (J–V) scans, B) transient photocurrent density–time (J–t), C) EIS measurements at 0 V vs. Ag/AgCl bias in a three-electrode system containing 0.1m Na2S and 0.02m Na2SO3 (pH 11.5) as the electrolyte under solar light irradiation for a) 0 mm, b) 5mm, c) 10 mm, and d) 15 mm Al2O3-coated CdS/Zr:Fe2O3 heterojunctions, d) photocurrent density vs. irradiation time for 30 min of bare and 10 mm Al2O3 over the CdS/Zr:Fe2O3 heterojunction.
표
A) Linear sweep voltammetric (J–V) scans, B) transient photocurrent density–time (J–t), C) EIS measurements at 0 V vs. Ag/AgCl bias in a three-electrode system containing 0.1m Na2S and 0.02m Na2SO3 (pH 11.5) as the electrolyte under solar light irradiation for a) 0 mm, b) 5mm, c) 10 mm, and d) 15 mm Al2O3-coated CdS/Zr:Fe2O3 heterojunctions, d) photocurrent density vs. irradiation time for 30 min of bare and 10 mm Al2O3 over the CdS/Zr:Fe2O3 heterojunction.
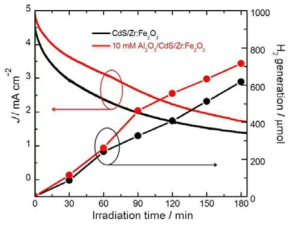 표
Photocurrent stability and hydrogen generation vs. irradiation time plots of uncoated CdS/Zr:Fe2O3 and 10 mm Al2O3-coated CdS/Zr:Fe2O3 heterojunction photoanodes recorded in an aqueous solution containing 0.1m Na2S and 0.02m Na2SO3 (pH&11.5) under one sun illumination at a potential of 0 V vs. Ag/AgCl.
표
Photocurrent stability and hydrogen generation vs. irradiation time plots of uncoated CdS/Zr:Fe2O3 and 10 mm Al2O3-coated CdS/Zr:Fe2O3 heterojunction photoanodes recorded in an aqueous solution containing 0.1m Na2S and 0.02m Na2SO3 (pH&11.5) under one sun illumination at a potential of 0 V vs. Ag/AgCl.
 표
(a) –(c) are 2D XRD images of the pristine(black dot), Ex-situ (blue dot), and In-situ (red dot) Sn-doped hematite photoanodes annealed at 800˚C, respectively. (d)-(e) The circular line cuts passing the hematite (110) and (104) peaks in which zero degree corresponds to the surface normal direction.
표
(a) –(c) are 2D XRD images of the pristine(black dot), Ex-situ (blue dot), and In-situ (red dot) Sn-doped hematite photoanodes annealed at 800˚C, respectively. (d)-(e) The circular line cuts passing the hematite (110) and (104) peaks in which zero degree corresponds to the surface normal direction.
 표
Cross-sectional scanning electron microscopic images of α-Fe2O3 photoanodes prepared by (a)one-step and (b) two-step annealing method.The textured regionis represented by a dark-field scanning electron microscopy images at a magnification of 150k and corresponds to the elemental maps of Fe, Sn, and O. The samples were partly melted due to the high energy of FIB during the sample preparation. (c) Schematic illustration of growth mechanism during the one-and two-step annealing processes.
표
Cross-sectional scanning electron microscopic images of α-Fe2O3 photoanodes prepared by (a)one-step and (b) two-step annealing method.The textured regionis represented by a dark-field scanning electron microscopy images at a magnification of 150k and corresponds to the elemental maps of Fe, Sn, and O. The samples were partly melted due to the high energy of FIB during the sample preparation. (c) Schematic illustration of growth mechanism during the one-and two-step annealing processes.
 표
(a) Jph variation of one-step annealed hematite photoanodes measured at 1.23 VRHE in 1M NaOH for different annealing times. For comparison, Jph value at 1.23V RHE of two-step-annealed α-Fe2O3 photoanode prepared by similar higher temperature annealing condition is shown. (b) J–V curves of one- and two-step annealed hematite photoanodes with similar high temperature annealing at 800 ˚C for 13.5 min. The error bars represent the standard deviations in PEC measurements of the independently prepared series of hematite photoanodes. Light illumination, 1sun; scan rate, 50mVs-1.
표
(a) Jph variation of one-step annealed hematite photoanodes measured at 1.23 VRHE in 1M NaOH for different annealing times. For comparison, Jph value at 1.23V RHE of two-step-annealed α-Fe2O3 photoanode prepared by similar higher temperature annealing condition is shown. (b) J–V curves of one- and two-step annealed hematite photoanodes with similar high temperature annealing at 800 ˚C for 13.5 min. The error bars represent the standard deviations in PEC measurements of the independently prepared series of hematite photoanodes. Light illumination, 1sun; scan rate, 50mVs-1.
 표
FE-SEM images of photoanodes (a) without and (b) with a GO underlayer.(c) Raman spectra of FTO/GO, FTO/GO/β-FeOOH, and FTO/GO/α-Fe2O3 photoanodes sintered at 800˚C. (d) Synchrotron X-ray diffraction patterns of α -Fe2O3(marked in blue) hotoanodes on FTO substrates(remaining peaks) sintered at 800oC without (black line) and with a GO(red line) interlayer.(e) Williamson-Hall plots of the hematite peaks denoted as blue dots in (d). The dashed lines represent linear regressions.
표
FE-SEM images of photoanodes (a) without and (b) with a GO underlayer.(c) Raman spectra of FTO/GO, FTO/GO/β-FeOOH, and FTO/GO/α-Fe2O3 photoanodes sintered at 800˚C. (d) Synchrotron X-ray diffraction patterns of α -Fe2O3(marked in blue) hotoanodes on FTO substrates(remaining peaks) sintered at 800oC without (black line) and with a GO(red line) interlayer.(e) Williamson-Hall plots of the hematite peaks denoted as blue dots in (d). The dashed lines represent linear regressions.
 표
(a) Photocurrent-potential (J-V) curves and (b) Transient photocurrent measurement for the PEC water oxidation reaction with and without GO-based α-Fe2O3 photoanodes in FTO substrates sintered at 800˚C. 1M NaOH was used under 1sun standard illumination conditions and the inset shows the photocurrent densities of the hematite photoanodes with and without GO underlayers at 1.4V vs. RHE under 1 sun standard illumination conditions.
표
(a) Photocurrent-potential (J-V) curves and (b) Transient photocurrent measurement for the PEC water oxidation reaction with and without GO-based α-Fe2O3 photoanodes in FTO substrates sintered at 800˚C. 1M NaOH was used under 1sun standard illumination conditions and the inset shows the photocurrent densities of the hematite photoanodes with and without GO underlayers at 1.4V vs. RHE under 1 sun standard illumination conditions.
 표
Current density–voltage (J–V) curves of (a) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by LT-annealing at 550 1C, (b) pristine a-Fe2O3 photoanodes fabricated under different annealing conditions, (c) pristine and 1 mM Sn-doped a-Fe2O3 photoanodes fabricated at high temperature by two-step and one-step annealing methods, and (d) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by one-step HT-annealing at 800 1C. The tangent arrows pointing toward the X-axis gives the onset potential (Vonset) of all the a-Fe2O3 photoanodes.
표
Current density–voltage (J–V) curves of (a) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by LT-annealing at 550 1C, (b) pristine a-Fe2O3 photoanodes fabricated under different annealing conditions, (c) pristine and 1 mM Sn-doped a-Fe2O3 photoanodes fabricated at high temperature by two-step and one-step annealing methods, and (d) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by one-step HT-annealing at 800 1C. The tangent arrows pointing toward the X-axis gives the onset potential (Vonset) of all the a-Fe2O3 photoanodes.
 표
Nyquist plots (real vs. imaginary impedance) of (a) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by LT-annealing at 550 1C, (b) pristine a-Fe2O3 photoanodes fabricated under different annealing conditions, (c) pristine and 1 mM Sn-doped a-Fe2O3 photoanodes fabricated at a high temperature by two-step and one-step annealing methods, and (d) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by one-step HT-annealing at 800 1C. The EIS data were obtained at a bias potential of 1.23 VRHE under 1 sun illumination in 1 M NaOH. The discrete symbols and the solid lines/curves in the plots represent the experimental data and the results of fitting, respectively. The inset of (a) shows the equivalent electrochemical circuit used for fitting all the Nyquist plots.
표
Nyquist plots (real vs. imaginary impedance) of (a) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by LT-annealing at 550 1C, (b) pristine a-Fe2O3 photoanodes fabricated under different annealing conditions, (c) pristine and 1 mM Sn-doped a-Fe2O3 photoanodes fabricated at a high temperature by two-step and one-step annealing methods, and (d) pristine and Sn-doped a-Fe2O3 photoanodes fabricated by one-step HT-annealing at 800 1C. The EIS data were obtained at a bias potential of 1.23 VRHE under 1 sun illumination in 1 M NaOH. The discrete symbols and the solid lines/curves in the plots represent the experimental data and the results of fitting, respectively. The inset of (a) shows the equivalent electrochemical circuit used for fitting all the Nyquist plots.
 표
(a) Jph variation at 1.23 VRHE for PRED-treated Fe2O3/Fe photoanodes prepared at 800˚C with different annealing times. The error bars represent the standard deviations in J–V measurements. The inset of (a) shows the J–V curves of bare and PRED-treated Fe2O3/Fe photoanodes annealed at 800˚C for 6 min, and (b) shows the corresponding photo-stability curves. Magnified portions show the current-responses under chopped illumination (100 mW cm−2) at 1.23 VRHE in 1 M NaOH.
표
(a) Jph variation at 1.23 VRHE for PRED-treated Fe2O3/Fe photoanodes prepared at 800˚C with different annealing times. The error bars represent the standard deviations in J–V measurements. The inset of (a) shows the J–V curves of bare and PRED-treated Fe2O3/Fe photoanodes annealed at 800˚C for 6 min, and (b) shows the corresponding photo-stability curves. Magnified portions show the current-responses under chopped illumination (100 mW cm−2) at 1.23 VRHE in 1 M NaOH.
 표
A. Schematic of charge transfer mechanism in metal oxide coated ZT photoanode. B. 28. Evolution of H2 and photocurrent density as a function of time measured at 0.1 V vs. Ag/AgCl using SZT as a photoelectrode under 100mW cm−2 light illumination in a three-electrode configuration, Inset shows a schematic of the photoelectrochemical hydrogen generation.
표
A. Schematic of charge transfer mechanism in metal oxide coated ZT photoanode. B. 28. Evolution of H2 and photocurrent density as a function of time measured at 0.1 V vs. Ag/AgCl using SZT as a photoelectrode under 100mW cm−2 light illumination in a three-electrode configuration, Inset shows a schematic of the photoelectrochemical hydrogen generation.
 표
A. Schematic mechanism of the cascading electron and hole transfer in the ternar CdS/ZnIn2S4/TiO2 heterojunction system. B. Evolution of H2 and photocurrent density as a function of time form the CdS/ZnIn2S4/TiO2 photoanode under 100 mW cm2 light illumination. The inset shows a picture of the simple photoelectrochemical cell setup used for hydrogen generation (WE: working electrode, CE: counter electrode, and RE: reference electrode).
표
A. Schematic mechanism of the cascading electron and hole transfer in the ternar CdS/ZnIn2S4/TiO2 heterojunction system. B. Evolution of H2 and photocurrent density as a function of time form the CdS/ZnIn2S4/TiO2 photoanode under 100 mW cm2 light illumination. The inset shows a picture of the simple photoelectrochemical cell setup used for hydrogen generation (WE: working electrode, CE: counter electrode, and RE: reference electrode).
 표
(a) Photocurrent density – potential characteristics, (b) long-term photostability and transient photocurrent responses, (c) EIS plots of CdS/ZnIn2S4/TiO2 photoelectrodes prepared with different nickel precursor concentrations(5, 10, 15 and 25 mM). Inset shows the equivalent circuit, and (d) Photocurrent stability measurement of a CdS/ZnIn2S4/TiO2 and 10mM Ni(OH)2/CdS/ZnIn2S4/TiO2 heterojunctionphotoelectrodesat an applied potential of 0.1 V (vs Ag/AgCl) for 1 h.
표
(a) Photocurrent density – potential characteristics, (b) long-term photostability and transient photocurrent responses, (c) EIS plots of CdS/ZnIn2S4/TiO2 photoelectrodes prepared with different nickel precursor concentrations(5, 10, 15 and 25 mM). Inset shows the equivalent circuit, and (d) Photocurrent stability measurement of a CdS/ZnIn2S4/TiO2 and 10mM Ni(OH)2/CdS/ZnIn2S4/TiO2 heterojunctionphotoelectrodesat an applied potential of 0.1 V (vs Ag/AgCl) for 1 h.
 표
A) Linear sweep voltammetric (J–V) scans, B) transient photocurrent density–time (J–t), C) EIS measurements at 0 V vs. Ag/AgCl bias in a three-electrode system containing 0.1m Na2S and 0.02m Na2SO3 (pH 11.5) as the electrolyte under solar light irradiation for a) 0 mm, b) 5mm, c) 10 mm, and d) 15 mm Al2O3-coated CdS/Zr:Fe2O3 heterojunctions, d) photocurrent density vs. irradiation time for 30 min of bare and 10 mm Al2O3 over the CdS/Zr:Fe2O3 heterojunction.
표
A) Linear sweep voltammetric (J–V) scans, B) transient photocurrent density–time (J–t), C) EIS measurements at 0 V vs. Ag/AgCl bias in a three-electrode system containing 0.1m Na2S and 0.02m Na2SO3 (pH 11.5) as the electrolyte under solar light irradiation for a) 0 mm, b) 5mm, c) 10 mm, and d) 15 mm Al2O3-coated CdS/Zr:Fe2O3 heterojunctions, d) photocurrent density vs. irradiation time for 30 min of bare and 10 mm Al2O3 over the CdS/Zr:Fe2O3 heterojunction.
 표
Photocurrent stability and hydrogen generation vs. irradiation time plots of uncoated CdS/Zr:Fe2O3 and 10 mm Al2O3-coated CdS/Zr:Fe2O3 heterojunction photoanodes recorded in an aqueous solution containing 0.1m Na2S and 0.02m Na2SO3 (pH&11.5) under one sun illumination at a potential of 0 V vs. Ag/AgCl.
표
Photocurrent stability and hydrogen generation vs. irradiation time plots of uncoated CdS/Zr:Fe2O3 and 10 mm Al2O3-coated CdS/Zr:Fe2O3 heterojunction photoanodes recorded in an aqueous solution containing 0.1m Na2S and 0.02m Na2SO3 (pH&11.5) under one sun illumination at a potential of 0 V vs. Ag/AgCl.
| 과제명(ProjectTitle) : | - |
|---|---|
| 연구책임자(Manager) : | - |
| 과제기간(DetailSeriesProject) : | - |
| 총연구비 (DetailSeriesProject) : | - |
| 키워드(keyword) : | - |
| 과제수행기간(LeadAgency) : | - |
| 연구목표(Goal) : | - |
| 연구내용(Abstract) : | - |
| 기대효과(Effect) : | - |
Copyright KISTI. All Rights Reserved.

※ AI-Helper는 부적절한 답변을 할 수 있습니다.