최소 단어 이상 선택하여야 합니다.
최대 10 단어까지만 선택 가능합니다.
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
NTIS 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
DataON 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Edison 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Kafe 바로가기
| 주관연구기관 | 국립식량과학원 National Institute of Crop Science |
|---|---|
| 보고서유형 | 최종보고서 |
| 발행국가 | 대한민국 |
| 언어 | 한국어 |
| 발행년월 | 2013-01 |
| 과제시작연도 | 2012 |
| 주관부처 | 농촌진흥청 Rural Development Administration(RDA) |
| 등록번호 | TRKO201300013904 |
| 과제고유번호 | 1395025540 |
| 사업명 | 국책기술개발 |
| DB 구축일자 | 2013-07-29 |
| DOI | https://doi.org/10.23000/TRKO201300013904 |
Ⅲ. 연구개발의 내용 및 범위
○ 농업부산물이용 바이오에탄올 생산 기술 개발
○ 셀룰로오스계 농업부산물 종류별 특성분석
○ 셀룰로오스계 농업부산물의 고효율 에탄올 전환 미생물 이용기술 개발
○ 우수 셀룰로오스 당화효소 최적생산조건 확립
○ 셀룰로스계 농업부산물 대량확보 방안 및 수거시스템 구축
○ 곰팡이 유래 셀룰로스계 농업부산물 분해 및 당화용효소 고농도 생산
○ 농업부산물로부터 Z. mobilis와 혐기성 섬유소 분해효소 생산 미생물 혼합/단계적배양을 통한 효율적인 에탄올 생산 공정 개발
Ⅲ. 연구개발의 내용 및 범위
○ 농업부산물이용 바이오에탄올 생산 기술 개발
○ 셀룰로오스계 농업부산물 종류별 특성분석
○ 셀룰로오스계 농업부산물의 고효율 에탄올 전환 미생물 이용기술 개발
○ 우수 셀룰로오스 당화효소 최적생산조건 확립
○ 셀룰로스계 농업부산물 대량확보 방안 및 수거시스템 구축
○ 곰팡이 유래 셀룰로스계 농업부산물 분해 및 당화용효소 고농도 생산
○ 농업부산물로부터 Z. mobilis와 혐기성 섬유소 분해효소 생산 미생물 혼합/단계적배양을 통한 효율적인 에탄올 생산 공정 개발
○ 효모유전공학을 이용한 통합공정발효균주 개발
The importance of bioenergy production has increased because of deficit of fossil energy and for the preparation of climate change. Collection system of raw materials for bioenergy production is required. As the agricultural byproducts is dispersed widely, reasonable collection system is required co
The importance of bioenergy production has increased because of deficit of fossil energy and for the preparation of climate change. Collection system of raw materials for bioenergy production is required. As the agricultural byproducts is dispersed widely, reasonable collection system is required considering geographical dispersion, road network, transporting facility and collection cost.
We analyzed the generation and utilization of cellulose agricultural byproducts according to season and region, the collection cost of agricultural byproducts, and how to decide the biorefinery place and optimize the collection system using GIS.
Forty one microorganisms, expected to be cellulolytic or xylanolytic microorganisms were isolated from various environmental samples, such as soil, miscanthus residue, horse manure, compost, humus, etc. These strains were screened at 35℃ by using plate count atgar containing 1% (w/v) carboxymethyl cellulose (CMC) or xylan (from the beech wood) instead of glucose. All of them were rod form and gram positive strains. Among the strains, both D1 and E2 microbial strains, which have high cellulase and xylanase activities, were characterized and identified as Bacillus subtilis by analysis of 16S rDNA sequence and biochemical studies, and named as B. subtilis D1 and E2, respectively. The maximum cellulase activities of D1 and E2 were 38.5 and 30.9 IU. Optimum temperature and pH of D1 and E2 for the cellulase activity were 50℃, pH 6 and 7, respectively. The cellulase activity was strongly inhibited by Al+++ and Zn++. The maximum xylanase activities of D1 and E2 were 467.3 and 310.5 IU. Optimum temperature and pH of D1 and E2 for the cellulase activity were 60℃, pH 10 and 11, respectively. The cellulase gene was cloned from each B. subtilis D1 and E2 genomic DNA by polymerase chain reaction (PCR). The amplified PCR products were ligated with the T&A cloning vector system and the constructed plasmids were transformed into E. coli DH5α. The sequence analysis of the insert DNAs revealed the identification of a 1,499-bp region containing cellulase open reading frame. According to cellulase gene sequence analysis, B. subtilis D1 had gene sequence similarity of 98% with B. subtilis strain AH18 cellulase gene (EF070194.1) and B. subtilis I15-endo-1,4-beta-glucanase (FJ464332.1). In case of B. subtilis E2, it had gene sequence similarity of 99% with B. subtilis strain AH18 cellulase gene (EF070194.1) and B.subtilis CMCase gene (D01057.1). The recombinant plasmid (including B. subtilis D1 cellulase gene) which was ligated with pET-28a(+) vector was expressed in E. coli BL21 and the expressed fusion protein was analyzed by SDS-PAGE. A new specific band with molecular weight of about 50 kDa was obtained from cellular extract of E. coli BL21 harboring cellulase gene. IPTG concentration and induction time were optimized for expression of cellulase gene. Optimal IPTG concentration and induction time 0.1 mM and 1 hr, respectively.
 표
Determination of molecular mass of purified YlLac by a) SDS-PAGE of laccase (M-Marker, L1-Crude protein, L2-DEAE cellulose purification, L3-Biogel Hiload 16/60 Superdex 200 chromatography), b) Zymogram activity and glycoprotein staining of purified laccase enzyme with native PAGE (L1-ABTS, L2-2,6-DMP, L3-glycoprotein staining). c) Determination of the native molecular mass of YlLac by gel filtration chromatography on a Sephacryl S-300 high-resolution column.
표
Determination of molecular mass of purified YlLac by a) SDS-PAGE of laccase (M-Marker, L1-Crude protein, L2-DEAE cellulose purification, L3-Biogel Hiload 16/60 Superdex 200 chromatography), b) Zymogram activity and glycoprotein staining of purified laccase enzyme with native PAGE (L1-ABTS, L2-2,6-DMP, L3-glycoprotein staining). c) Determination of the native molecular mass of YlLac by gel filtration chromatography on a Sephacryl S-300 high-resolution column.
 표
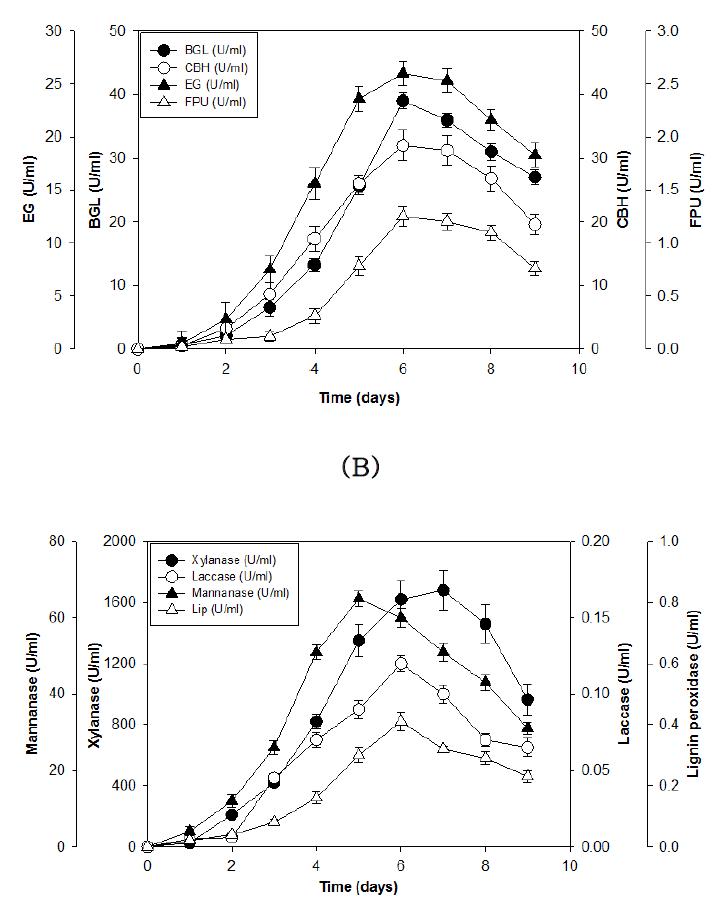
(A) Time course of total cellulase activity of (FPU) BGL, CBH and EG production in 70-L fermenter by P . adiposa SKU0714. () BGL activity; (О) CBH activity; (▲) EG activity; (Δ) FPU activity. (B) Time course of xylanase, laccase, mannanase, and lignin peroxidase activities of P . adiposa SKU0714. () Xylanase activity; (О) Laccase activity; (▲) Mannanase activity; (Δ) Lignin peroxidase activity.
표
(A) Time course of total cellulase activity of (FPU) BGL, CBH and EG production in 70-L fermenter by P . adiposa SKU0714. () BGL activity; (О) CBH activity; (▲) EG activity; (Δ) FPU activity. (B) Time course of xylanase, laccase, mannanase, and lignin peroxidase activities of P . adiposa SKU0714. () Xylanase activity; (О) Laccase activity; (▲) Mannanase activity; (Δ) Lignin peroxidase activity.
 표
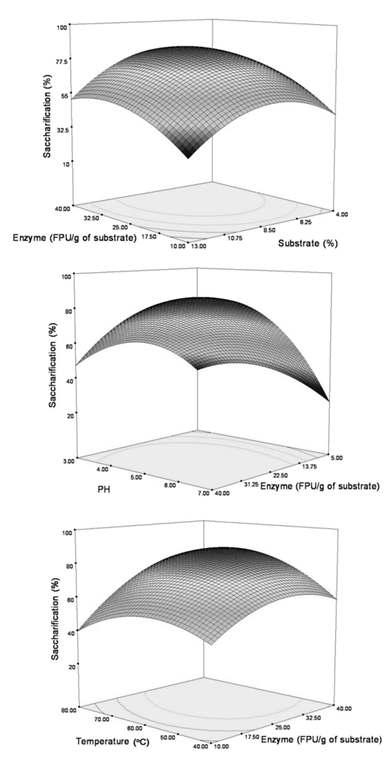
Response surface plots showing the relationships between independent variables in the conversion of cellulose to reducing sugars: (A) relationship between substrate and enzyme concentration; (B) relationship between enzyme concentration and pH; (C) relationship between enzyme concentration and temperature.
표
Response surface plots showing the relationships between independent variables in the conversion of cellulose to reducing sugars: (A) relationship between substrate and enzyme concentration; (B) relationship between enzyme concentration and pH; (C) relationship between enzyme concentration and temperature.
 표
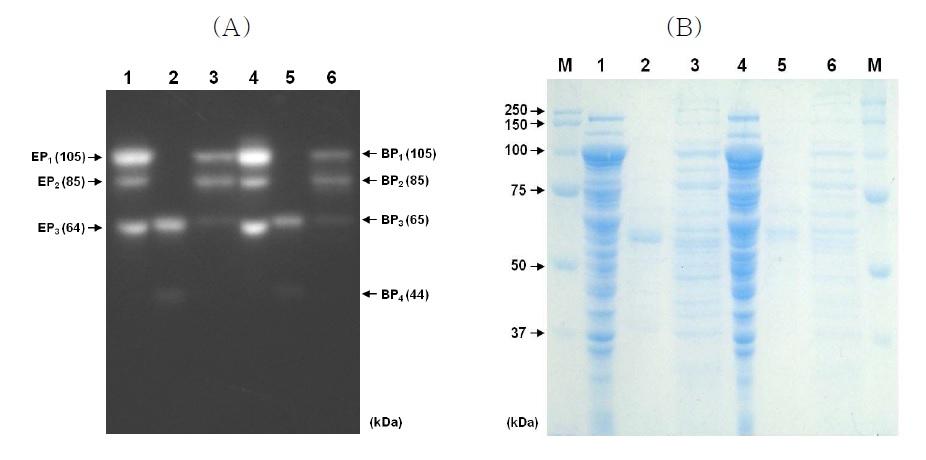
Activity staining (a) and protein staining (b) after SDS-PAGE of the xylanase produced by E. coli BL21(DE3) (pCX33), B. subtilis WB700 (pJX33) and B. subtilis WB800 (pJX33). Lanes 1 and 4, crude extract of E. coli BL21(DE3) (pCX33); 2 and 5, concentrated supernatant of B. subtilis WB700 (pJX33); 3 and 6,concentrated supernatant of B. subtilis WB800 (pJX33).
표
Activity staining (a) and protein staining (b) after SDS-PAGE of the xylanase produced by E. coli BL21(DE3) (pCX33), B. subtilis WB700 (pJX33) and B. subtilis WB800 (pJX33). Lanes 1 and 4, crude extract of E. coli BL21(DE3) (pCX33); 2 and 5, concentrated supernatant of B. subtilis WB700 (pJX33); 3 and 6,concentrated supernatant of B. subtilis WB800 (pJX33).
 표
Determination of molecular mass of purified YlLac by a) SDS-PAGE of laccase (M-Marker, L1-Crude protein, L2-DEAE cellulose purification, L3-Biogel Hiload 16/60 Superdex 200 chromatography), b) Zymogram activity and glycoprotein staining of purified laccase enzyme with native PAGE (L1-ABTS, L2-2,6-DMP, L3-glycoprotein staining). c) Determination of the native molecular mass of YlLac by gel filtration chromatography on a Sephacryl S-300 high-resolution column.
표
Determination of molecular mass of purified YlLac by a) SDS-PAGE of laccase (M-Marker, L1-Crude protein, L2-DEAE cellulose purification, L3-Biogel Hiload 16/60 Superdex 200 chromatography), b) Zymogram activity and glycoprotein staining of purified laccase enzyme with native PAGE (L1-ABTS, L2-2,6-DMP, L3-glycoprotein staining). c) Determination of the native molecular mass of YlLac by gel filtration chromatography on a Sephacryl S-300 high-resolution column.
 표
(A) Time course of total cellulase activity of (FPU) BGL, CBH and EG production in 70-L fermenter by P . adiposa SKU0714. () BGL activity; (О) CBH activity; (▲) EG activity; (Δ) FPU activity. (B) Time course of xylanase, laccase, mannanase, and lignin peroxidase activities of P . adiposa SKU0714. () Xylanase activity; (О) Laccase activity; (▲) Mannanase activity; (Δ) Lignin peroxidase activity.
표
(A) Time course of total cellulase activity of (FPU) BGL, CBH and EG production in 70-L fermenter by P . adiposa SKU0714. () BGL activity; (О) CBH activity; (▲) EG activity; (Δ) FPU activity. (B) Time course of xylanase, laccase, mannanase, and lignin peroxidase activities of P . adiposa SKU0714. () Xylanase activity; (О) Laccase activity; (▲) Mannanase activity; (Δ) Lignin peroxidase activity.
 표
Response surface plots showing the relationships between independent variables in the conversion of cellulose to reducing sugars: (A) relationship between substrate and enzyme concentration; (B) relationship between enzyme concentration and pH; (C) relationship between enzyme concentration and temperature.
표
Response surface plots showing the relationships between independent variables in the conversion of cellulose to reducing sugars: (A) relationship between substrate and enzyme concentration; (B) relationship between enzyme concentration and pH; (C) relationship between enzyme concentration and temperature.
 표
Activity staining (a) and protein staining (b) after SDS-PAGE of the xylanase produced by E. coli BL21(DE3) (pCX33), B. subtilis WB700 (pJX33) and B. subtilis WB800 (pJX33). Lanes 1 and 4, crude extract of E. coli BL21(DE3) (pCX33); 2 and 5, concentrated supernatant of B. subtilis WB700 (pJX33); 3 and 6,concentrated supernatant of B. subtilis WB800 (pJX33).
표
Activity staining (a) and protein staining (b) after SDS-PAGE of the xylanase produced by E. coli BL21(DE3) (pCX33), B. subtilis WB700 (pJX33) and B. subtilis WB800 (pJX33). Lanes 1 and 4, crude extract of E. coli BL21(DE3) (pCX33); 2 and 5, concentrated supernatant of B. subtilis WB700 (pJX33); 3 and 6,concentrated supernatant of B. subtilis WB800 (pJX33).
| 과제명(ProjectTitle) : | - |
|---|---|
| 연구책임자(Manager) : | - |
| 과제기간(DetailSeriesProject) : | - |
| 총연구비 (DetailSeriesProject) : | - |
| 키워드(keyword) : | - |
| 과제수행기간(LeadAgency) : | - |
| 연구목표(Goal) : | - |
| 연구내용(Abstract) : | - |
| 기대효과(Effect) : | - |
Copyright KISTI. All Rights Reserved.

※ AI-Helper는 부적절한 답변을 할 수 있습니다.