최소 단어 이상 선택하여야 합니다.
최대 10 단어까지만 선택 가능합니다.
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
NTIS 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
DataON 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Edison 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Kafe 바로가기
| 주관연구기관 | 전남대학교 산학협력단 Chonnam National University |
|---|---|
| 보고서유형 | 2단계보고서 |
| 발행국가 | 대한민국 |
| 언어 | 한국어 |
| 발행년월 | 2014-03 |
| 과제시작연도 | 2013 |
| 주관부처 | 미래창조과학부 Ministry of Science, ICT and Future Planning |
| 과제관리전문기관 | 한국연구재단 National Research Foundation of Korea |
| 등록번호 | TRKO201500002491 |
| 과제고유번호 | 1711003654 |
| 사업명 | 바이오·의료기술개발 |
| DB 구축일자 | 2015-05-16 |
| 키워드 | 바이러스.치료제.동물모델.효능.안전성.검증.산업화.Virus.Medicine.Animal model.Efficacy.Safety.Evaluation.Industrialization. |
| DOI | https://doi.org/10.23000/TRKO201500002491 |
1차년도에서는 로타바이러스 치료후보물질의 효능 및 안전성 검증을 위해 동물모델을 확립하였으며, 치료후보물질인 EC를 다양한 농도별로 로타바이러스성 장염이 유발된 동물모델에 투여한 결과 항로타바이러스 효능은 있었지만 실험동물이 폐사하는 등 안전성에 문제가 있었음. 2차년도에서는 SW-100 및 JSC를 로타바이러스성 장염이 유발된 동물모델에 다양한 농도별로 각각 혹은 혼합물 형태로 투여한 결과 SW-100은 치료효과가 없었고, JSC는 로타바이러스성 장염의 개선효과가 있었으며, 이들 혼합물은 로타바이러스성 장염을 완벽히 치료하였음.
1차년도에서는 로타바이러스 치료후보물질의 효능 및 안전성 검증을 위해 동물모델을 확립하였으며, 치료후보물질인 EC를 다양한 농도별로 로타바이러스성 장염이 유발된 동물모델에 투여한 결과 항로타바이러스 효능은 있었지만 실험동물이 폐사하는 등 안전성에 문제가 있었음. 2차년도에서는 SW-100 및 JSC를 로타바이러스성 장염이 유발된 동물모델에 다양한 농도별로 각각 혹은 혼합물 형태로 투여한 결과 SW-100은 치료효과가 없었고, JSC는 로타바이러스성 장염의 개선효과가 있었으며, 이들 혼합물은 로타바이러스성 장염을 완벽히 치료하였음. 3차년도에서는 메탄올 추출물 형태의 KW-200 및 KW-200 분획물을 동물모델에 투여한 결과 완벽한 치료효과가 있었으며 랫트 및 자돈에서 안전성이 높았음. 하지만 KW-200의 주요 성분인 glycyrrhizin은 그 효능이 KW-200에 비해 낮았음. 제4차년도에서는 압착 생추출물 형태의 KW-200을 동물모델에 투여한 결과 완벽한 치료 효과와 랫트와 자돈에서 안전성이 높았음. ST-100 및 JSC 혼합제제에 대한 효능 및 안전성을 재검증한 결과, 제2차년도의 결과와 유사하게 매우 높았음. 또한 메탄올 추출물 형태인 KW-200에 대한 효능을 재검증한 결과 제3차년도와 유사하게 매우 높았음. 제5차년도에서는 KW-200의 송아지에서의 로타바이러스, 코로나바이러스, 로타바이러스와 코로나바이러스 혼합감염에 대한 효능을 검증한 결과 설사 완화효과가 높았음. 이상의 결과 2차년도-5차년도에서 검증된 로타바이러스 및 코로나바이러스 치료 후보물질은 돼지 로타바이러스성 장염 및 소 로타바이러스 및 코로나바이러스의 치료제로 가축에서 바로 활용할 수 있을 것이며, 인간의 로타바이러스 치료제 전임상 연구결과로 사용할 수 있을 것임.
Ⅳ. Results
▣ 1st year
● Development of rotavirus animal model: Before evaluating the efficacy and safety of antirotavirus drug candidates, we established rotavirus animal models with piglets and calves. Col-D piglets obtained from sows by hysterectomy were inoculated with a porcine rotavirus s
Ⅳ. Results
▣ 1st year
● Development of rotavirus animal model: Before evaluating the efficacy and safety of antirotavirus drug candidates, we established rotavirus animal models with piglets and calves. Col-D piglets obtained from sows by hysterectomy were inoculated with a porcine rotavirus strain. Piglets inoculated experimentally with porcine rotavirus showed severe diarrhea, fecal virus shedding, and histological changes in intestine. Col-D calves inoculated experimentally with bovine rotavirus also exhibited severe diarrhea, fecal virus shedding, and histological changes in intestine. These results concluded that the piglets and calves can be useful animal models for evaluating the efficacy and safety of antirotavirus drug candidates.
● Evaluation of efficacy and safety of antirotavirus drug candidate, EC: To evaluate the efficacy and safety of antirotavirus drug candidate, EC, the piglets after induction of rotavirus diarrhea were treated with various concentration of EC drug candidate. Even the piglets treated with drug candidate showed the alleviation of clinical signs, histological changes of intestine, fecal virus shedding, and antigen localization, during experimental period, some piglets died. These results implied that this candidate had toxic effect to piglets so that could not be used for drug due to safety problem.
● Evaluation of virus-host interaction: To evaluate the virus-host interaction, microarray system was used. Significant changes (either increase or decrease) of host mRNA expression levels were detected in many genes from virus inoculated or drug-treated cells. These data indicate that the increase or decrease of host genes can be used as the points acted by drugs.
● Analysis of full-length 11 genomic segments of rotaviruses: In order to analyze full-length 11 genomic segments of rotaviruses to be used for establishing rotavirus animal model, eleven genomic segments of various rotavirus strains were sequenced after RT-PCR and 5' and 3' RACE. Various genotypes and mutations were detected, indicating that different therapeutics might be applied for each different strain even in animals or humans.
▣ 2nd year
● Evaluation of efficacy and safety of SW-100 in animal model as an antirotavirus drug candidate: The piglets treated with SW-100 alone did not show any improvement of diarrhea score, fecal virus shedding, and small intestinal lesion in comparison with those of virus-inoculated and mock-treated piglets. However, this compound did not cause any toxic effects to piglets including the abnormal clinical signs, mortality, histological changes in any organ or tissue, etc.
● Evaluation of efficacy and safety of JSC in animal model as an antirotavirus drug candidate: Dose-dependently, JSC treated groups showed gradual decrease of diarrhea score, and improvement of fecal virus shedding and small intestinal lesion, but did not completely cure diarrhea. Moreover, this candidate did not cause any toxic effects to piglets including the abnormal clinical signs, mortality, histological changes in any organ or tissue, etc.
● Evaluation of efficacy and safety, and synergistic effect by combination therapy of various mixtures of JSC and SW-100 in animal model as an antirotavirus drug candidate: Combination therapy of JSC and SW-100 completely cured diarrhea at post-inoculation day 8. Furthermore, it markedly improved the small intestinal lesion and fecal virus shedding. Moreover, this combination therapy did not cause any toxic effects to piglets including the abnormal clinical signs, mortality, histological changes in any organ or tissue, etc. These data indicate that the combination therapy of JSC and SW-100 can be used to livestock and humans as an anti-rotavirus drug.
▣ 3rd year
● Evaluation of efficacy and safety of methanol-extracted KW-200 in animal model as an antirotavirus drug candidate: At 100 mg/ml and 200 mg/ml concentrations of KW-200, fecal consistency, fecal virus shedding and histological changes of intestinal lesions did not improved in piglets after rotaviral diarrhea induction. Interestingly, 400 mg/ml of KW-200 halted diarrhea at post-inoculation day 8 and markedly improved small intestinal lesion score and fecal virus shedding. Moreover, this candidate did not cause any toxic effects to piglets including the abnormal clinical signs, mortality, histological changes in any organ or tissue, etc.
● Evaluation of anti-proimflammatory efficacy of methanol-extracted KW-200: Decrease levels for mRNA expression for IL-8, IL-10, TNF-α, IFN-β, IFN-γ, NF-kB, p38, and JNK were accounted for those piglets that have received treatment in comparison to the virus-inoculated and mock-treated group.
● Overall, methanol-extracted KW-200 exerted anti-rotavirus effects as well as anti-proimflammatory efficacy. These data implied that KW-200 can be used to livestock and humans as an anti-rotavirus drug.
● Safety assessment of methanol-extracted KW-200 in mice: Compared to mock-treated group, orally or intraperitoneally administrated groups with various concentrations of methanol extracted KW-200 showed any abnormality in body weight, organ weight, blood cells as well as blood chemistries including HCT, MCH, MCHC, MCV, PLT, etc. These data indicate that methanol-extracted KW-200 were safe in mice.
● Safety assessment of methanol-extracted KW-200 in piglets: Compared to mock-treated group, orally or intraperitoneally administrated groups with various concentrations of methanol extracted KW-200 showed any abnormality in body weight, organ weight, blood cells as well as blood chemistries including HCT, MCH, MCHC, MCV, PLT, etc. These data indicate that methanol-extracted KW-200 were safe in piglets.
● Evaluation of efficacy and safety of KW-200 fraction in animal model as an anti-rotavirus drug candidate: At 10 mg/ml and 25 mg/ml concentrations of KW-200 fraction, fecal consistency, fecal virus shedding and histological changes of intestinal lesions did not improved in piglets after rotaviral diarrhea induction. Interestingly, 50 mg/ml of KW-200 fraction halted diarrhea at post-inoculation day 8 and improved small intestinal lesion score and fecal virus shedding.
● Evaluation of anti-proimflammatory efficacy of KW-200 fraction: Decrease levels for mRNA expression for NF-kB, IL8, IFN-β and TNF-α were accounted for those piglets that have received treatment in comparison to the virus-inoculated and mock-treated group. This candidate did not cause any toxic effects to piglets including the abnormal clinical signs, mortality, histological changes in any organ or tissue, etc.
● Evaluation of efficacy and safety of glycyrrhizin in animal model as an anti-rotavirus drug candidate: Compared to mock-treated and virus-inoculated group, glycyrrhizin-administrated group showed any improvement on diarrhea score, fecal virus shedding and histopathological intestinal lesion. These data indicate that glycyrrhizin might not be drug candidate for curring rotavirus enteritis.
● Development of a new animal model with G9 bearing porcine rotavirus strain: Development of a new animal model with G9 bearing porcine rotavirus strain: In order to develop rotavirus animal model with G9 bearing strain, G9P[7] and G9P[23] strains isolated in swine were inoculated into Col-D piglets produced in sows by hysterotomy, and intestinal and extra-intestinal pathogenicity were evaluated. The piglets inoculated with each strain showed typical diarrhea and histological changes in the intestinal and systemic organs, indicating these strains to be valuable to evaluate the efficacy and safety of antirotavirus drug candidates not only for animals but also humans.
▣ 4th year
● Evaluation of efficacy and safety of compression-extracted KW-200 in animal model as an antirotavirus drug candidate: At 100 mg/ml and 200 mg/ml concentrations of KW-200, fecal consistency, fecal virus shedding and histological changes of intestinal lesions did not improved in piglets after rotaviral diarrhea induction. Interestingly, 400 and 600 mg/ml of KW-200 cured diarrhea at post-inoculation day 8 and markedly improved small intestinal lesion score and fecal virus shedding. Moreover, 800 mg/kg shortened cure date to post-inoculation day 6 and improved small intestinal lesion score and fecal virus shedding more than those by lower concentration. In additon, this candidate did not cause any toxic effects to piglets including the abnormal clinical signs, mortality, histological changes in any organ or tissue, etc.
● Evaluation of anti-proimflammatory efficacy of compression-extracted KW-200: Decrease levels for mRNA expression for IL-8, IL-10, TNF-α, IFN-β, IFN-γ, NF-kB, p38, and JNK were accounted for those piglets that have received treatment in comparison to the virus-inoculated and mock-treated group.
● Overall, compression-extracted KW-200 exerted stronger anti-rotavirus effects as well as anti-proimflammatory efficacy than those by methanol-extracted KW-200. These data implied that compression-extracted KW-200 can be used to livestock and humans as an anti-rotavirus drug.
● Safety assessment of compression-extracted KW-200 in mice: Compared to mock-treated group, orally or intraperitoneally administrated groups with various concentrations of compression- extracted KW-200 showed any abnormality in body weight, organ weight, blood cells as well as blood chemistries including HCT, MCH, MCHC, MCV, PLT, etc. These data indicate that like methanol-extracted KW-200, compression-extracted KW-200 were also safe in mice.
● Safety assessment of compression-extracted KW-200 in piglets: Compared to mock-treated group, orally or intraperitoneally administrated groups with various concentrations of compression extracted KW-200 showed any abnormality in body weight, organ weight, blood cells as well as blood chemistries including HCT, MCH, MCHC, MCV, PLT, etc. These data indicate that like methanol-extracted KW-200, compression-extracted KW-200 were safe in piglets.
● Reevaluation of efficacy and safety, and synergistic effect by combination therapy of various mixtures of JSC and SW-100 in animal model as an antirotavirus drug candidate: In order to address the points raised by reviewers, the efficacy and safety of anti-rotavirus drug candidate were reevaluated using Col-D piglets. Combination therapy of JSC and SW-100 completely cured diarrhea at post-inoculation day 8. Furthermore, it markedly improved the small intestinal lesion and fecal virus shedding. Moreover, this combination therapy did not cause any toxic effects to piglets including the abnormal clinical signs, mortality, histological changes in any organ or tissue, etc. These data indicate that like the data shown in the part of 2nd year results, the combination therapy of JSC and SW-100 can be used to livestock and humans as an anti-rotavirus drug.
● Safety assessment of various mixtures of JSC and SW-100 in mice: Compared to mock-treated group, orally or intraperitoneally administrated groups with various concentrations of JSC and SW-100 mixtures showed any abnormality in body weight, organ weight, blood cells as well as blood chemistries including HCT, MCH, MCHC, MCV, PLT, etc. These data indicate that various combinations of JSC and SW-100 were safe in mice.
● Safety assessment of various mixtures of JSC and SW-100 in piglets: Compared to mock-treated group, orally or intraperitoneally administrated groups with various concentrations of JSC and SW-100 mixtures showed any abnormality in body weight, organ weight, blood cells as well as blood chemistries including HCT, MCH, MCHC, MCV, PLT, etc. These data indicate that various combinations of JSC and SW-100 were safe in piglets.
● Reevaluation of efficacy and safety of methanol-extracted KW-200 in animal model as an antirotavirus drug candidate: In order to address the points raised by reviewers, the efficacy and safety of anti-rotavirus drug candidate were reevaluated using Col-D piglets. At 100 mg/ml and 200 mg/ml concentrations of KW-200, fecal consistency, fecal virus shedding and histological changes of intestinal lesions did not improved in piglets after rotaviral diarrhea induction. Interestingly, 400 mg/ml of KW-200 halted diarrhea at post-inoculation day 8 and markedly improved small intestinal lesion score and fecal virus shedding. Moreover, this candidate did not cause any toxic effects to piglets including the abnormal clinical signs, mortality, histological changes in any organ or tissue, etc.
● Revaluation of anti-proimflammatory efficacy of methanol-extracted KW-200: In order to address the points raised by reviewers, the efficacy and safety of anti-rotavirus drug candidate were reevaluated using Col-D piglets. Decrease levels for mRNA expression for IL-8, IL-10, TNF-α, IFN-β, IFN-γ, NF-kB, p38, and JNK were accounted for those piglets that have received treatment in comparison to the virus-inoculated and mock-treated group.
● Overall, the anti-rotavirus efficacy and safety of methanol-extracted KW-200 were also demonstrated in the second trial. Moreover, anti-proimflammatory efficacy was also proved. These data implied that KW-200 can be used to livestock and humans as an anti-rotavirus drug.
▣ 5th year
● Evaluation of efficacy of methanol-extracted KW-200 in calf model as an anti-rotavirus drug candidate: Bovine rotavirus is one of the major enteric pathogens in calves worldwide. To evaluate whether methanol-extracted KW-200 exerted anti-rotavirus efficacy, Col-D calves were administered with methanol-extracted KW-200 after experimental induction of rotavirus diarrhea. Compared to mock-treated but virus-inoculated calves, KW-200 alleviated rotavirus diarrhea at post-inoculation day 7. Moreover, KW-200 obtunded fecal virus shedding and histopathological intestinal lesions. These data indicated that like cure effects in piglets, KW-200 can be used as drug to calves for curing the rotavirus diarrhea.
● Evaluation of efficacy of methanol-extracted KW-200 in calf model as an anti-coronavirus drug candidate: Among the enteric pathogens for calf diarrhea, bovine coronavirus is one of the major enteric pathogens in calves worldwide. To evaluate whether methanol-extracted KW-200 exerted anti-coronavirus efficacy, Col-D calves were administered with methanol-extracted KW-200 after experimental induction of coronavirus diarrhea. Compared to mock-treated but virus-inoculated calves, KW-200 alleviated coronavirus diarrhea at post-inoculation day 7. Moreover, KW-200 obtunded fecal virus shedding and histopathological intestinal lesions. These data indicated that like cure effects for rotavirus diarrhea in calves, KW-200 can be used as drug to calves for curing the coronavirus diarrhea.
● Evaluation of efficacy of methanol-extracted KW-200 in calf model as an anti-rotavirus and anti-coronavirus drug candidate: In the field calves, calf diarrhea can be caused by two more pathogens in particular with coinfection with rotavirus and coronavirus. Therefore, anti-viral medicine should exert its effects to more than 2 viral pathogens. To fulfill this condition, Col-D calves were co-infected with bovine rotavirus and coronavirus. At the point of diarrhea induced by both enteric viruses, methanol-extracted KW-200 were administrated. Accordingly, KW-200 alleviated diarrhea score as well as fecal virus shedding and intestinal lesion score. These data indicate that KW-200 can be used as drug for curing diarrhea due to coinfection of both rotavirus and coronavirus.
● Development of a new animal model with G5P[7] bearing porcine and bovine rotavirus strains: To choose the rotavirus strain which can induce diarrhea to piglets and calves, G5P[7] bearing porcine and bovine rotavirus strains were selected and then inoculated into Col-D piglets and Col-D calves. Porcine G5P[7] strain only induced diarrhea and intestinal and extra-intestinal pathology in piglets but not in calves. However, bovine G5P[7] strain induced diarrhea and intestinal and extra-intestinal pathology in piglets and calves. These data indicate that bovine G5P[7] rotavirus strain is useful for evaluating efficacy and safety of anti-rotavirus drug candidate in piglets and calves.
 표
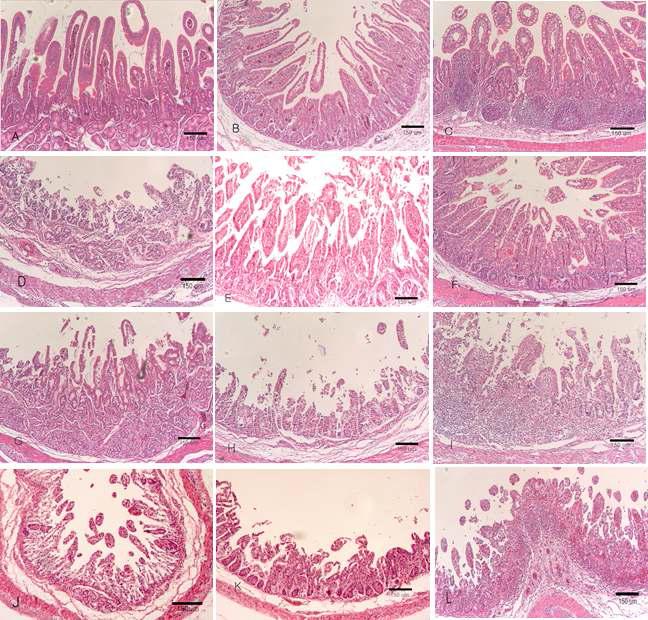
Duodenum, Jejunum, Ileum: A-C, Piglets inoculated with G5P7 rotavirus (5 x 104/ml virus titer). A Moderate changes in the duodenum. B Moderate changes in the jejunum. C Moderate changes in the ileum. D-F, G5P7 (5 x 10/ml5) strain inoculated piglets. D Severe villi atrophy and villi hyperplasia in the duodenum. E. Moderate villi atrophy and villi hyperplasia in the jejunum. F. Moderate villi atrophy and crypt hyperplasia in the ileum. G-I, G5P7(5x106/ml) inoculated piglets. G Severe villi atrophy and villi hyperplasia in the duodenum. H Severe villi atrophy and villi hyperplasia in the jejunum. I Severe villi atrophy and crypt hyperplasia in the ileum. J-L, G5P7(5x107/ml) inoculated piglets. J Severe villi atrophy and crypt hyperplasia in the duodenum. K Severe villi atrophy and crypt hyperplasia in the jejunum. L Marked villi atrophy and crypt hyperplasia in the ileum. Hematoxylin and eosin stain. Bar: A -L, 150㎛.
표
Duodenum, Jejunum, Ileum: A-C, Piglets inoculated with G5P7 rotavirus (5 x 104/ml virus titer). A Moderate changes in the duodenum. B Moderate changes in the jejunum. C Moderate changes in the ileum. D-F, G5P7 (5 x 10/ml5) strain inoculated piglets. D Severe villi atrophy and villi hyperplasia in the duodenum. E. Moderate villi atrophy and villi hyperplasia in the jejunum. F. Moderate villi atrophy and crypt hyperplasia in the ileum. G-I, G5P7(5x106/ml) inoculated piglets. G Severe villi atrophy and villi hyperplasia in the duodenum. H Severe villi atrophy and villi hyperplasia in the jejunum. I Severe villi atrophy and crypt hyperplasia in the ileum. J-L, G5P7(5x107/ml) inoculated piglets. J Severe villi atrophy and crypt hyperplasia in the duodenum. K Severe villi atrophy and crypt hyperplasia in the jejunum. L Marked villi atrophy and crypt hyperplasia in the ileum. Hematoxylin and eosin stain. Bar: A -L, 150㎛.
![Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G8P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy and crypt hyperplasia in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛. Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G8P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy and crypt hyperplasia in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_47_image_8.jpg) 표
Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G8P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy and crypt hyperplasia in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.
표
Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G8P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy and crypt hyperplasia in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.
![Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G5P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy ,crypt hyperplasia and lyphoid cell infiltratioin in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛. Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G5P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy ,crypt hyperplasia and lyphoid cell infiltratioin in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_48_image_9.jpg) 표
Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G5P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy ,crypt hyperplasia and lyphoid cell infiltratioin in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.
표
Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G5P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy ,crypt hyperplasia and lyphoid cell infiltratioin in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.
![Antigen localization of rotaviruses; calf A-C Heterologous G5P[7] strain-inoculated calf. A -B. Lung sampled at PID(Post-inoculation day) 7 from calf inoculated with the heterolotous bovine rotavirus G5P[7] strain. C. Liver sampled at PID 7 from calf inoculated with the geterologous bovine rotavirus G5P[7]. A-B, 100㎛ C 200㎛. Antigen localization of rotaviruses; calf A-C Heterologous G5P[7] strain-inoculated calf. A -B. Lung sampled at PID(Post-inoculation day) 7 from calf inoculated with the heterolotous bovine rotavirus G5P[7] strain. C. Liver sampled at PID 7 from calf inoculated with the geterologous bovine rotavirus G5P[7]. A-B, 100㎛ C 200㎛.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_49_image_10.jpg) 표
Antigen localization of rotaviruses; calf A-C Heterologous G5P[7] strain-inoculated calf. A -B. Lung sampled at PID(Post-inoculation day) 7 from calf inoculated with the heterolotous bovine rotavirus G5P[7] strain. C. Liver sampled at PID 7 from calf inoculated with the geterologous bovine rotavirus G5P[7]. A-B, 100㎛ C 200㎛.
표
Antigen localization of rotaviruses; calf A-C Heterologous G5P[7] strain-inoculated calf. A -B. Lung sampled at PID(Post-inoculation day) 7 from calf inoculated with the heterolotous bovine rotavirus G5P[7] strain. C. Liver sampled at PID 7 from calf inoculated with the geterologous bovine rotavirus G5P[7]. A-B, 100㎛ C 200㎛.
![Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain. D. Duodenum showing severe villi atrophy and crypt cell hyperplasia. E. Severe villi atrophy and crypt hyperplasia in the jejunum. F. Ileum showing severe villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC after diarrhea induction by a G5P[7] strain showing slight villi atrophy and crypt hyperplasia. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. H&E stain. Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain. D. Duodenum showing severe villi atrophy and crypt cell hyperplasia. E. Severe villi atrophy and crypt hyperplasia in the jejunum. F. Ileum showing severe villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC after diarrhea induction by a G5P[7] strain showing slight villi atrophy and crypt hyperplasia. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. H&E stain.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_89_image_53.jpg) 표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain. D. Duodenum showing severe villi atrophy and crypt cell hyperplasia. E. Severe villi atrophy and crypt hyperplasia in the jejunum. F. Ileum showing severe villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC after diarrhea induction by a G5P[7] strain showing slight villi atrophy and crypt hyperplasia. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. H&E stain.
표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain. D. Duodenum showing severe villi atrophy and crypt cell hyperplasia. E. Severe villi atrophy and crypt hyperplasia in the jejunum. F. Ileum showing severe villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC after diarrhea induction by a G5P[7] strain showing slight villi atrophy and crypt hyperplasia. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. H&E stain.
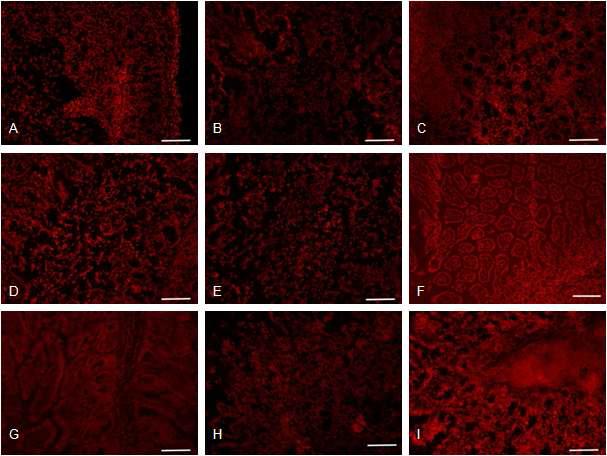
![Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 200 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. (M-O) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus. Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 200 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. (M-O) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_91_image_55.jpg) 표
Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 200 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. (M-O) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
표
Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 200 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. (M-O) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
![Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain showing severe villi atrophy and crypt hyperplasia. (D-F) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain exhibiting slight to mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC showing slight to mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC revealing slight to mild villi atrophy and crypt hyperplasia. Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain showing severe villi atrophy and crypt hyperplasia. (D-F) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain exhibiting slight to mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC showing slight to mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC revealing slight to mild villi atrophy and crypt hyperplasia.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_98_image_60.jpg) 표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain showing severe villi atrophy and crypt hyperplasia. (D-F) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain exhibiting slight to mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC showing slight to mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC revealing slight to mild villi atrophy and crypt hyperplasia.
표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain showing severe villi atrophy and crypt hyperplasia. (D-F) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain exhibiting slight to mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC showing slight to mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC revealing slight to mild villi atrophy and crypt hyperplasia.
![Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus. Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_100_image_62.jpg) 표
Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
표
Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
![Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain exhibiting severe atrophy and fusion of villi. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in saline after diarrhea induction by the inoculation of a porcine rotavirus G5P[7] strain showing severe atrophy and fusion of villi, crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets starved for 1 day before treatment of ST-100 in saline revealing severe atrophy and fusion of villi. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in milk showing severe villi atrophy and fusion. H&E stain. Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain exhibiting severe atrophy and fusion of villi. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in saline after diarrhea induction by the inoculation of a porcine rotavirus G5P[7] strain showing severe atrophy and fusion of villi, crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets starved for 1 day before treatment of ST-100 in saline revealing severe atrophy and fusion of villi. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in milk showing severe villi atrophy and fusion. H&E stain.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_109_image_67.jpg) 표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain exhibiting severe atrophy and fusion of villi. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in saline after diarrhea induction by the inoculation of a porcine rotavirus G5P[7] strain showing severe atrophy and fusion of villi, crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets starved for 1 day before treatment of ST-100 in saline revealing severe atrophy and fusion of villi. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in milk showing severe villi atrophy and fusion. H&E stain.
표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain exhibiting severe atrophy and fusion of villi. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in saline after diarrhea induction by the inoculation of a porcine rotavirus G5P[7] strain showing severe atrophy and fusion of villi, crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets starved for 1 day before treatment of ST-100 in saline revealing severe atrophy and fusion of villi. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in milk showing severe villi atrophy and fusion. H&E stain.
 표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. See normal and histopathological changes of small intestine in the upper figures. (A-C) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 100 mg/ml JSC and 3 g ST-100 showing mild villi atrophy and crypt hyperplasia. (E-F) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 200 mg/ml JSC and 3 g ST-100 exhibiting mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 400 mg/ml JSC and 3 g ST-100 revealing slight villi atrophy and crypt hyperplasia. H&E stain.
표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. See normal and histopathological changes of small intestine in the upper figures. (A-C) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 100 mg/ml JSC and 3 g ST-100 showing mild villi atrophy and crypt hyperplasia. (E-F) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 200 mg/ml JSC and 3 g ST-100 exhibiting mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 400 mg/ml JSC and 3 g ST-100 revealing slight villi atrophy and crypt hyperplasia. H&E stain.
 표
Antigen distribution in the duodenum, jejunum, ileum. See antigen appearance in the small intestine sampled from normal and virus-inoculated piglets, respectively, in the upper figures. (A-C) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 100 mg/ml JSC and 3 g ST-100 revealing negative reactions in the villi epithelium for rotavirus antigen. (D-F) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 200 mg/ml JSC and 3 g ST-100 exhibiting negative reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 400 mg/ml JSC and 3 g ST-100 having negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
표
Antigen distribution in the duodenum, jejunum, ileum. See antigen appearance in the small intestine sampled from normal and virus-inoculated piglets, respectively, in the upper figures. (A-C) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 100 mg/ml JSC and 3 g ST-100 revealing negative reactions in the villi epithelium for rotavirus antigen. (D-F) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 200 mg/ml JSC and 3 g ST-100 exhibiting negative reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 400 mg/ml JSC and 3 g ST-100 having negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
![Experimental design for evaluating anti-rotavirus effects of methanol-extracted KW-200. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg KW-200-treated, group IV was virus-inoculated and 200 mg KW-200-treated, and group V was virus-inoculated and 400 mg/mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for evaluating anti-rotavirus effects of methanol-extracted KW-200. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg KW-200-treated, group IV was virus-inoculated and 200 mg KW-200-treated, and group V was virus-inoculated and 400 mg/mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_121_image_78.jpg) 표
Experimental design for evaluating anti-rotavirus effects of methanol-extracted KW-200. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg KW-200-treated, group IV was virus-inoculated and 200 mg KW-200-treated, and group V was virus-inoculated and 400 mg/mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for evaluating anti-rotavirus effects of methanol-extracted KW-200. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg KW-200-treated, group IV was virus-inoculated and 200 mg KW-200-treated, and group V was virus-inoculated and 400 mg/mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
 표
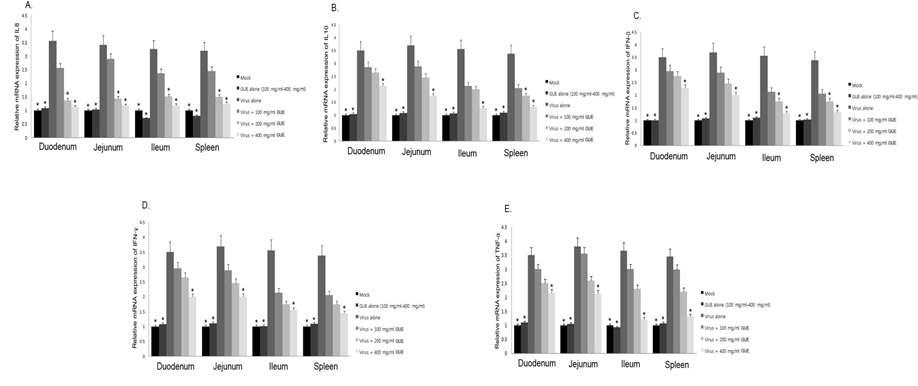
Influence of KW-200 on rotavirus-induced expression of IL8, IL10, IFN-β, IFN-γ, and TNF-α in colostrums-deprived piglets. Comparison of mRNA expression levels of IL8, IL10, IFN-β, IFN-γ, and TNF-α with those of β-actin in each experimental group was determined by SYBR Green Real-time RT-PCR. Values are means ± S.D. (n=3) (*p< 0.05).
표
Influence of KW-200 on rotavirus-induced expression of IL8, IL10, IFN-β, IFN-γ, and TNF-α in colostrums-deprived piglets. Comparison of mRNA expression levels of IL8, IL10, IFN-β, IFN-γ, and TNF-α with those of β-actin in each experimental group was determined by SYBR Green Real-time RT-PCR. Values are means ± S.D. (n=3) (*p< 0.05).
![Experimental design for evaluating anti-rotavirus effects of KW-200 fraction. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 10 mg KW-200 fraction-treated, group IV was virus-inoculated and 25 mg KW-200 fraction-treated, and group V was virus-inoculated, 50 mg/mg KW-200 fraction-treated. KW-200 fraction-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for evaluating anti-rotavirus effects of KW-200 fraction. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 10 mg KW-200 fraction-treated, group IV was virus-inoculated and 25 mg KW-200 fraction-treated, and group V was virus-inoculated, 50 mg/mg KW-200 fraction-treated. KW-200 fraction-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_134_image_93.jpg) 표
Experimental design for evaluating anti-rotavirus effects of KW-200 fraction. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 10 mg KW-200 fraction-treated, group IV was virus-inoculated and 25 mg KW-200 fraction-treated, and group V was virus-inoculated, 50 mg/mg KW-200 fraction-treated. KW-200 fraction-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for evaluating anti-rotavirus effects of KW-200 fraction. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 10 mg KW-200 fraction-treated, group IV was virus-inoculated and 25 mg KW-200 fraction-treated, and group V was virus-inoculated, 50 mg/mg KW-200 fraction-treated. KW-200 fraction-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
 표
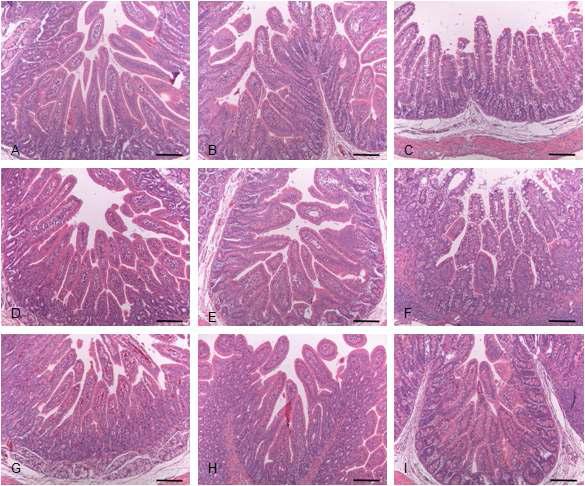
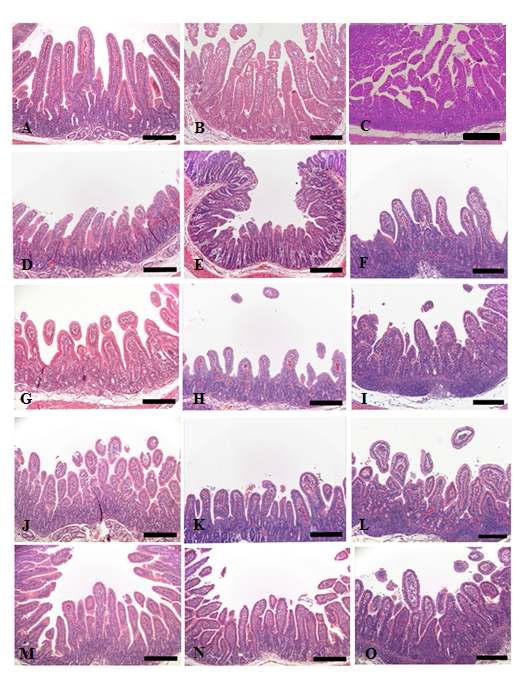
Histopathological changes of small intestine sampled from control and KW-200 fraction-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 10 mg/ml KW-200 fraction exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 25 mg/ml KW-200 fraction reveals slight improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). (M-O) Piglet treated with 50 mg/ml KW-200 shows moderate restored villi and crypt in the duodenum (M), jejunum (N) and ileum (O). Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and KW-200 fraction-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 10 mg/ml KW-200 fraction exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 25 mg/ml KW-200 fraction reveals slight improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). (M-O) Piglet treated with 50 mg/ml KW-200 shows moderate restored villi and crypt in the duodenum (M), jejunum (N) and ileum (O). Bars = 200 μm.
 표
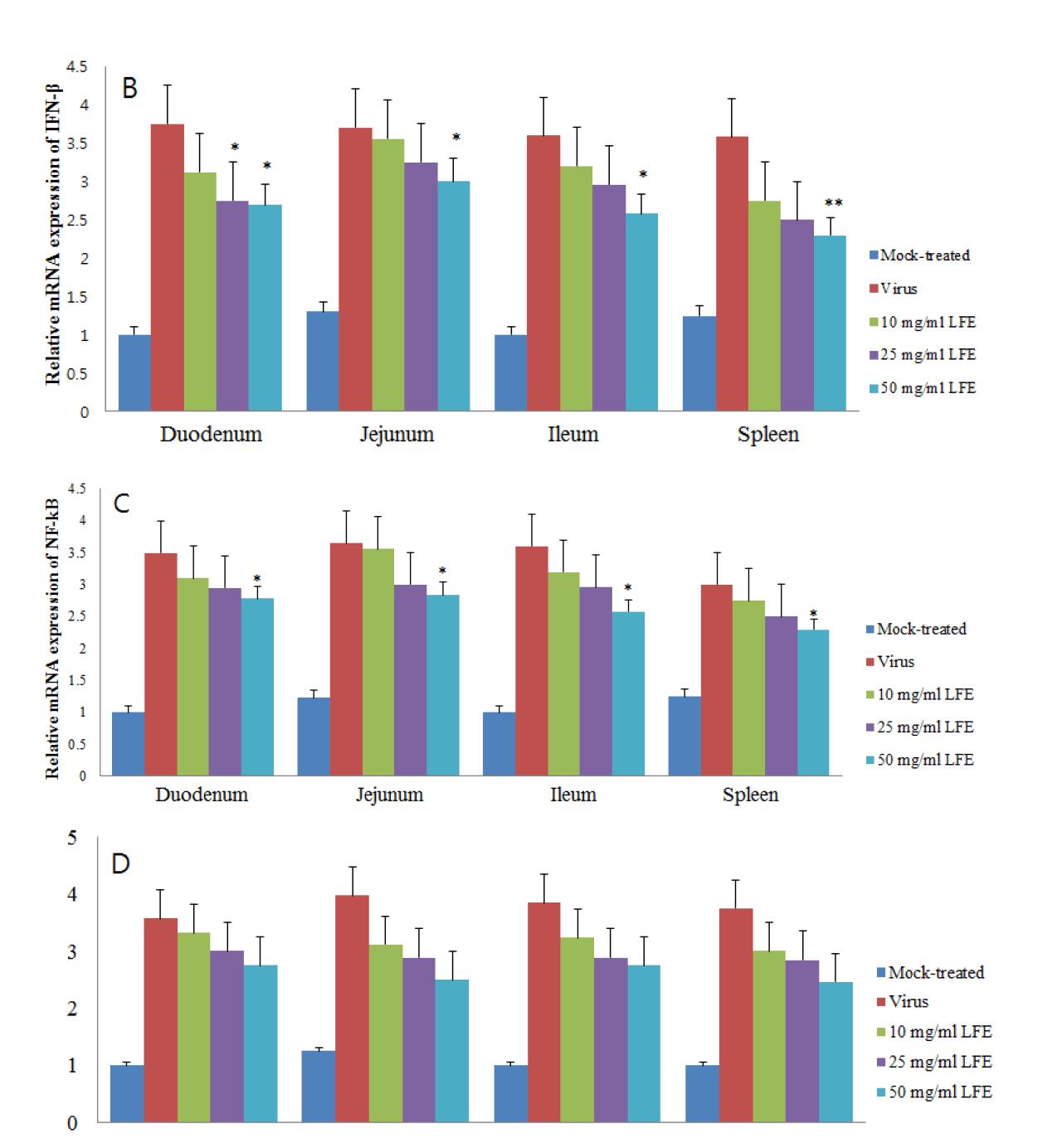
Influence of KW-200 fraction on rotavirus-induced expression of TNF-α (A), IFN-β (B), NF-κB (C), and IL8 (D) in colostrums-deprived piglets. Comparison of mRNA expression levels of TNF-α (A), IFN-β (B), NF-κB (C), and IL8 (D) with those of β-actin in each experimental group was determined by SYBR Green Real-time RT-PCR.
표
Influence of KW-200 fraction on rotavirus-induced expression of TNF-α (A), IFN-β (B), NF-κB (C), and IL8 (D) in colostrums-deprived piglets. Comparison of mRNA expression levels of TNF-α (A), IFN-β (B), NF-κB (C), and IL8 (D) with those of β-actin in each experimental group was determined by SYBR Green Real-time RT-PCR.
![Experimental design for evaluating anti-rotavirus effects of glycyrrhizin. Experimental animals were randomly allocated into 4 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg glycyrrhizin-treated, and group IV was virus-inoculated and 200 mg glycyrrhizin-treated. Glycyrrhizin-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for evaluating anti-rotavirus effects of glycyrrhizin. Experimental animals were randomly allocated into 4 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg glycyrrhizin-treated, and group IV was virus-inoculated and 200 mg glycyrrhizin-treated. Glycyrrhizin-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_140_image_98.jpg) 표
Experimental design for evaluating anti-rotavirus effects of glycyrrhizin. Experimental animals were randomly allocated into 4 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg glycyrrhizin-treated, and group IV was virus-inoculated and 200 mg glycyrrhizin-treated. Glycyrrhizin-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for evaluating anti-rotavirus effects of glycyrrhizin. Experimental animals were randomly allocated into 4 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg glycyrrhizin-treated, and group IV was virus-inoculated and 200 mg glycyrrhizin-treated. Glycyrrhizin-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
 표
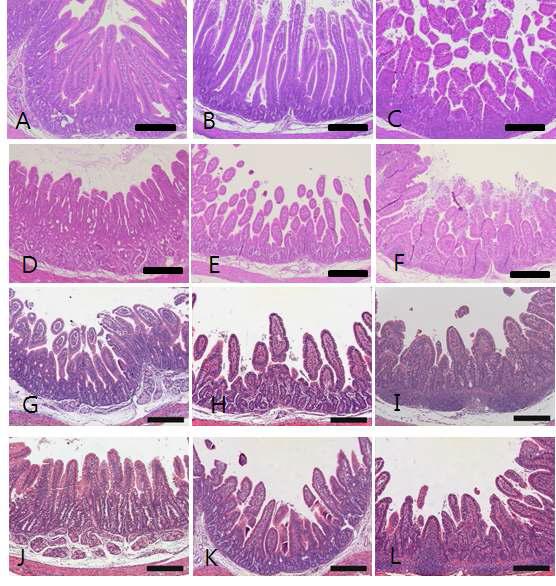
Histopathological changes of small intestine sampled from control and glycyrrhizin-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 100 mg/ml glycyrrhizin exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 200 mg/ml glycyrrhizin reveals no improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and glycyrrhizin-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 100 mg/ml glycyrrhizin exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 200 mg/ml glycyrrhizin reveals no improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
![Histopathological changes in the small intestine of piglets inoculated with PRG942 (G9P[23]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-inoculated piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm. Histopathological changes in the small intestine of piglets inoculated with PRG942 (G9P[23]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-inoculated piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_147_image_105.jpg) 표
Histopathological changes in the small intestine of piglets inoculated with PRG942 (G9P[23]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-inoculated piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.
표
Histopathological changes in the small intestine of piglets inoculated with PRG942 (G9P[23]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-inoculated piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.
![Histopathological changes in the small intestine of piglets inoculated with PRG9121 (G9P[7]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-infected piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet infected by PRG9121 (G9P[7]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm. Histopathological changes in the small intestine of piglets inoculated with PRG9121 (G9P[7]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-infected piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet infected by PRG9121 (G9P[7]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_148_image_106.jpg) 표
Histopathological changes in the small intestine of piglets inoculated with PRG9121 (G9P[7]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-infected piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet infected by PRG9121 (G9P[7]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.
표
Histopathological changes in the small intestine of piglets inoculated with PRG9121 (G9P[7]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-infected piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet infected by PRG9121 (G9P[7]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.
![Distribution of RVA-positive cells of PRG942 (G9P[23]) and PRG9121 (G9P[7]) strains in the duodenum. Piglets inoculated with either chloroform-inactivated PRG942 (G9P[23]) strain (A) or PRG9121 (G9P[7]) strain (B) contained no RVA-positive cells in the villi of each duodenum. RVA-positive cells (arrows) were scattered in the villi of duodenum sampled from piglets inoculated with PRG942 (G9P[23]) strain (C) or PRG9121 (G9P[7]) strain (D) at DPI 3. Indirect immunofluorescence assay was performed with monoclonal antibody against the VP6 protein of strain OSU. Bars A. D = 100 μm. Distribution of RVA-positive cells of PRG942 (G9P[23]) and PRG9121 (G9P[7]) strains in the duodenum. Piglets inoculated with either chloroform-inactivated PRG942 (G9P[23]) strain (A) or PRG9121 (G9P[7]) strain (B) contained no RVA-positive cells in the villi of each duodenum. RVA-positive cells (arrows) were scattered in the villi of duodenum sampled from piglets inoculated with PRG942 (G9P[23]) strain (C) or PRG9121 (G9P[7]) strain (D) at DPI 3. Indirect immunofluorescence assay was performed with monoclonal antibody against the VP6 protein of strain OSU. Bars A. D = 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_149_image_107.jpg) 표
Distribution of RVA-positive cells of PRG942 (G9P[23]) and PRG9121 (G9P[7]) strains in the duodenum. Piglets inoculated with either chloroform-inactivated PRG942 (G9P[23]) strain (A) or PRG9121 (G9P[7]) strain (B) contained no RVA-positive cells in the villi of each duodenum. RVA-positive cells (arrows) were scattered in the villi of duodenum sampled from piglets inoculated with PRG942 (G9P[23]) strain (C) or PRG9121 (G9P[7]) strain (D) at DPI 3. Indirect immunofluorescence assay was performed with monoclonal antibody against the VP6 protein of strain OSU. Bars A. D = 100 μm.
표
Distribution of RVA-positive cells of PRG942 (G9P[23]) and PRG9121 (G9P[7]) strains in the duodenum. Piglets inoculated with either chloroform-inactivated PRG942 (G9P[23]) strain (A) or PRG9121 (G9P[7]) strain (B) contained no RVA-positive cells in the villi of each duodenum. RVA-positive cells (arrows) were scattered in the villi of duodenum sampled from piglets inoculated with PRG942 (G9P[23]) strain (C) or PRG9121 (G9P[7]) strain (D) at DPI 3. Indirect immunofluorescence assay was performed with monoclonal antibody against the VP6 protein of strain OSU. Bars A. D = 100 μm.
![Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG942 (G9P[23]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (D) Normal lung tissue sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissue sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigens were detected in a pneumocyte (arrowhead) and lymphoid cell (arrow) in the lung from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrow). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a hepatocyte (arrow) in the liver from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigens were detected in the epithelial cells (arrow) of the choroid plexus from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. Bars A- L = 100 μm. Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG942 (G9P[23]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (D) Normal lung tissue sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissue sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigens were detected in a pneumocyte (arrowhead) and lymphoid cell (arrow) in the lung from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrow). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a hepatocyte (arrow) in the liver from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigens were detected in the epithelial cells (arrow) of the choroid plexus from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. Bars A- L = 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_153_image_111.jpg) 표
Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG942 (G9P[23]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (D) Normal lung tissue sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissue sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigens were detected in a pneumocyte (arrowhead) and lymphoid cell (arrow) in the lung from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrow). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a hepatocyte (arrow) in the liver from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigens were detected in the epithelial cells (arrow) of the choroid plexus from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. Bars A- L = 100 μm.
표
Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG942 (G9P[23]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (D) Normal lung tissue sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissue sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigens were detected in a pneumocyte (arrowhead) and lymphoid cell (arrow) in the lung from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrow). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a hepatocyte (arrow) in the liver from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigens were detected in the epithelial cells (arrow) of the choroid plexus from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. Bars A- L = 100 μm.
![Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG9121 (G9P[7]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (D) Normal lung tissues sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissues sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigen was detected in a pneumocyte (arrow) in the lung from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrows). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a few hepatocytes (arrows) in the liver from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigen was detected in a lymphoid cell (arrow) in the choroid plexus from a piglet inoculated with PRG9121 (G9P[7]) strain. Bars A- L = 100 μm. Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG9121 (G9P[7]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (D) Normal lung tissues sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissues sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigen was detected in a pneumocyte (arrow) in the lung from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrows). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a few hepatocytes (arrows) in the liver from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigen was detected in a lymphoid cell (arrow) in the choroid plexus from a piglet inoculated with PRG9121 (G9P[7]) strain. Bars A- L = 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_154_image_112.jpg) 표
Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG9121 (G9P[7]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (D) Normal lung tissues sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissues sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigen was detected in a pneumocyte (arrow) in the lung from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrows). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a few hepatocytes (arrows) in the liver from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigen was detected in a lymphoid cell (arrow) in the choroid plexus from a piglet inoculated with PRG9121 (G9P[7]) strain. Bars A- L = 100 μm.
표
Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG9121 (G9P[7]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (D) Normal lung tissues sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissues sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigen was detected in a pneumocyte (arrow) in the lung from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrows). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a few hepatocytes (arrows) in the liver from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigen was detected in a lymphoid cell (arrow) in the choroid plexus from a piglet inoculated with PRG9121 (G9P[7]) strain. Bars A- L = 100 μm.
![Quantification of RVA RNA by SYBR Green real-time RT-PCR. Real-time RT-PCR was performed with the feces, serum, mesenteric lymph node (MLN), liver, lung, choroid plexus, and nasal swab sampled from piglets inoculated with porcine G9 RVA strain PRG942 (G9P[23]) (A) and PRG9121 (G9P[7]) (B). The geometric means of viral RNA copy number per mg of tissue are displayed. Quantification of RVA RNA by SYBR Green real-time RT-PCR. Real-time RT-PCR was performed with the feces, serum, mesenteric lymph node (MLN), liver, lung, choroid plexus, and nasal swab sampled from piglets inoculated with porcine G9 RVA strain PRG942 (G9P[23]) (A) and PRG9121 (G9P[7]) (B). The geometric means of viral RNA copy number per mg of tissue are displayed.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_155_image_113.jpg) 표
Quantification of RVA RNA by SYBR Green real-time RT-PCR. Real-time RT-PCR was performed with the feces, serum, mesenteric lymph node (MLN), liver, lung, choroid plexus, and nasal swab sampled from piglets inoculated with porcine G9 RVA strain PRG942 (G9P[23]) (A) and PRG9121 (G9P[7]) (B). The geometric means of viral RNA copy number per mg of tissue are displayed.
표
Quantification of RVA RNA by SYBR Green real-time RT-PCR. Real-time RT-PCR was performed with the feces, serum, mesenteric lymph node (MLN), liver, lung, choroid plexus, and nasal swab sampled from piglets inoculated with porcine G9 RVA strain PRG942 (G9P[23]) (A) and PRG9121 (G9P[7]) (B). The geometric means of viral RNA copy number per mg of tissue are displayed.
![Experimental design for evaluating anti-rotavirus effects of compression-extracted KW-200 immediately after harvesting. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 200 mg KW-200-treated, group IV was virus-inoculated and 400 mg KW-200-treated, gorup V was virus-inoculated and 600 mg KW-200-treated, and group VI was virus-inoculated and 800 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for evaluating anti-rotavirus effects of compression-extracted KW-200 immediately after harvesting. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 200 mg KW-200-treated, group IV was virus-inoculated and 400 mg KW-200-treated, gorup V was virus-inoculated and 600 mg KW-200-treated, and group VI was virus-inoculated and 800 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_156_image_114.jpg) 표
Experimental design for evaluating anti-rotavirus effects of compression-extracted KW-200 immediately after harvesting. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 200 mg KW-200-treated, group IV was virus-inoculated and 400 mg KW-200-treated, gorup V was virus-inoculated and 600 mg KW-200-treated, and group VI was virus-inoculated and 800 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for evaluating anti-rotavirus effects of compression-extracted KW-200 immediately after harvesting. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 200 mg KW-200-treated, group IV was virus-inoculated and 400 mg KW-200-treated, gorup V was virus-inoculated and 600 mg KW-200-treated, and group VI was virus-inoculated and 800 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
![Experimental design for reevaluating anti-rotavirus effects on the various mixtures of ST-100 and JSC. This study was performed to address the points raised by reviewers. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for reevaluating anti-rotavirus effects on the various mixtures of ST-100 and JSC. This study was performed to address the points raised by reviewers. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_169_image_127.jpg) 표
Experimental design for reevaluating anti-rotavirus effects on the various mixtures of ST-100 and JSC. This study was performed to address the points raised by reviewers. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for reevaluating anti-rotavirus effects on the various mixtures of ST-100 and JSC. This study was performed to address the points raised by reviewers. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
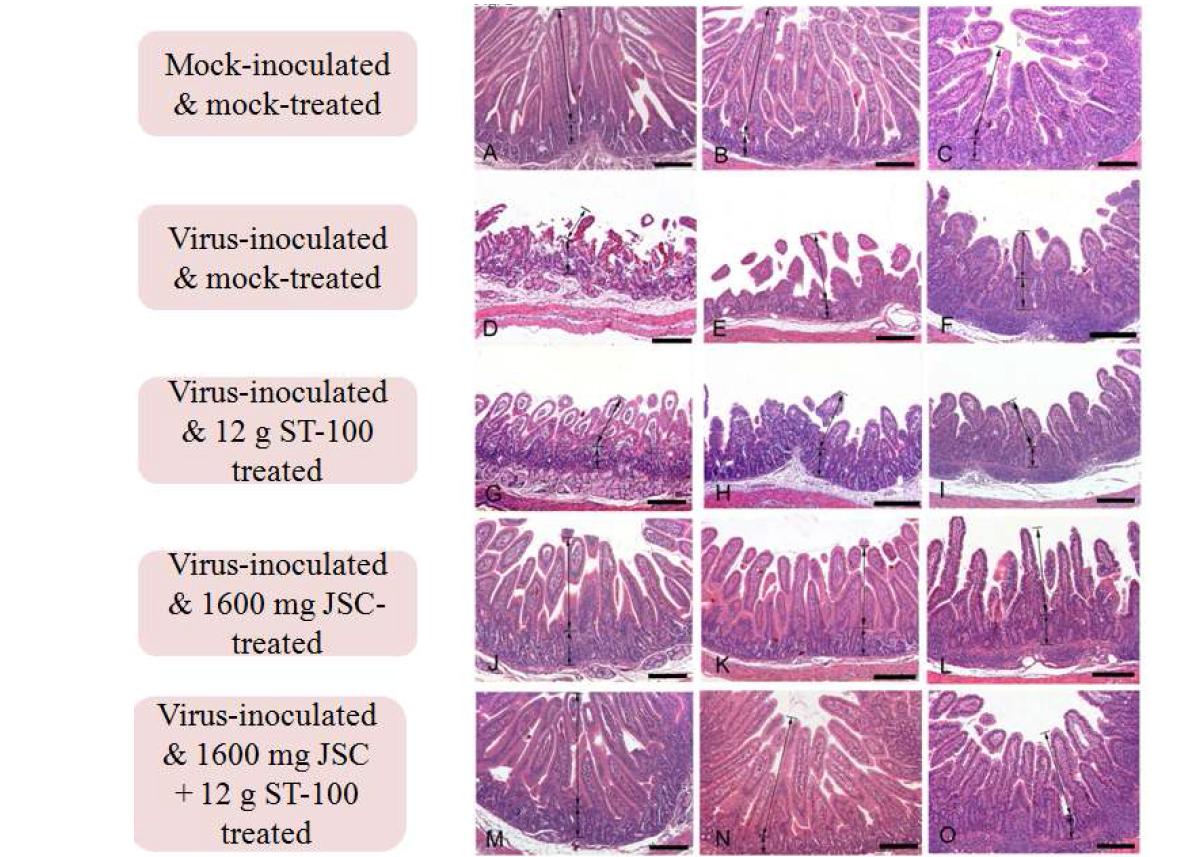 표
Histopathological changes of small intestine sampled from control and ST-100-JSC combination-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 12 g ST-100 exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 1600 mg JSC reveals moderate improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). (M-O) Piglet treated with 1600 mg JSC plus 12 g ST-100 shows markedly restored villi and crypt in the duodenum (M), jejunum (N) and ileum (O). (P-R) Piglet treated with 400 mg/ ml KW-200 shows unaltered duodenum (P), jejunum (Q) and ileum (R) with long and slender villi. Hematoxyline and eosin stain. Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and ST-100-JSC combination-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 12 g ST-100 exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 1600 mg JSC reveals moderate improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). (M-O) Piglet treated with 1600 mg JSC plus 12 g ST-100 shows markedly restored villi and crypt in the duodenum (M), jejunum (N) and ileum (O). (P-R) Piglet treated with 400 mg/ ml KW-200 shows unaltered duodenum (P), jejunum (Q) and ileum (R) with long and slender villi. Hematoxyline and eosin stain. Bars = 200 μm.
![Experimental design for reevaluating anti-rotavirus effects of KW-200 for addressing the points raised by reviewers. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 400 mg KW-200-treated, group IV was virus-inoculated and 800 mg KW-200-treated, and gorup V was virus-inoculated and 1600 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for reevaluating anti-rotavirus effects of KW-200 for addressing the points raised by reviewers. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 400 mg KW-200-treated, group IV was virus-inoculated and 800 mg KW-200-treated, and gorup V was virus-inoculated and 1600 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_180_image_139.jpg) 표
Experimental design for reevaluating anti-rotavirus effects of KW-200 for addressing the points raised by reviewers. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 400 mg KW-200-treated, group IV was virus-inoculated and 800 mg KW-200-treated, and gorup V was virus-inoculated and 1600 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for reevaluating anti-rotavirus effects of KW-200 for addressing the points raised by reviewers. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 400 mg KW-200-treated, group IV was virus-inoculated and 800 mg KW-200-treated, and gorup V was virus-inoculated and 1600 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
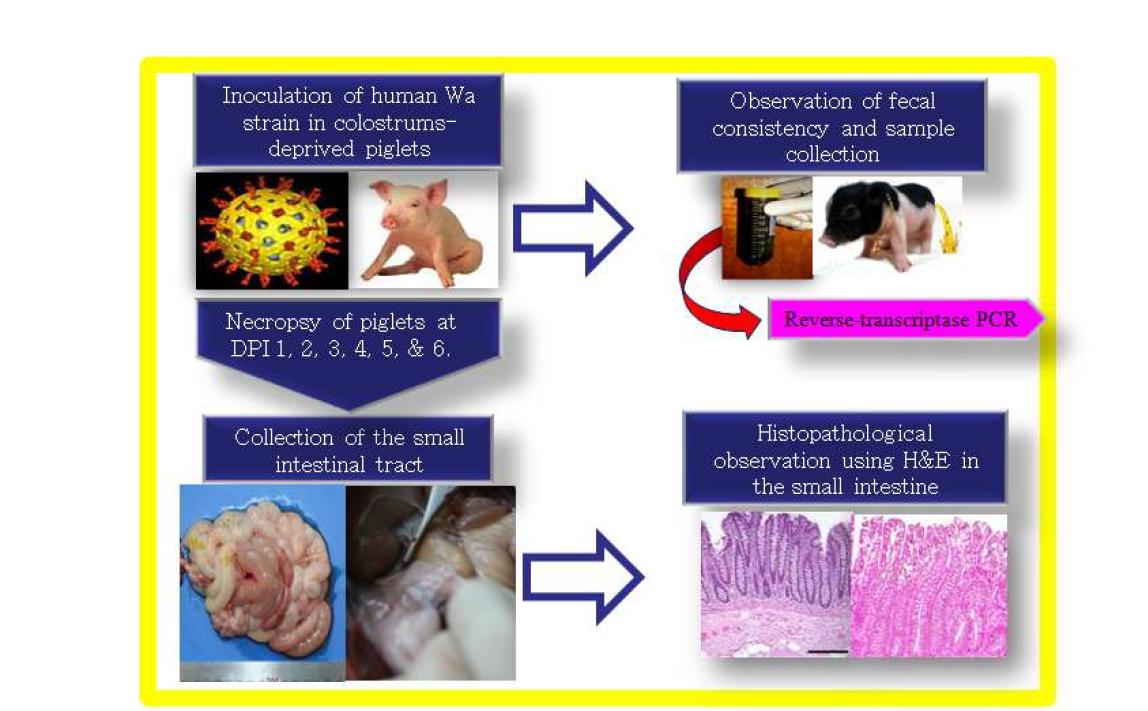 표
Experimental design for establishing human rotavirus experimental model. Piglets produced from sows by hysterectomy were kept in germ-free isolators. Experimental animals were inoculated with a human rotavirus strain Wa at 4 days old and euthanasied at day post-inoculation 1-6. Clinical signs including the diarrhea were monitored everyday and fecal specimens were sampled daily. All experimental animals. At necropsy, duodenum, jejunum and ileum were sampled for histopathological examination.
표
Experimental design for establishing human rotavirus experimental model. Piglets produced from sows by hysterectomy were kept in germ-free isolators. Experimental animals were inoculated with a human rotavirus strain Wa at 4 days old and euthanasied at day post-inoculation 1-6. Clinical signs including the diarrhea were monitored everyday and fecal specimens were sampled daily. All experimental animals. At necropsy, duodenum, jejunum and ileum were sampled for histopathological examination.
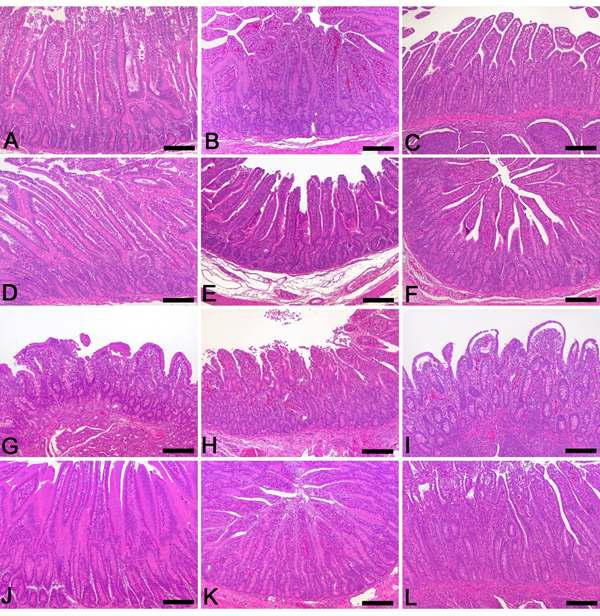 표
Histopathological changes of small intestine sampled from control and KW-200-treated groups. (A-C) Control calf shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Calf treated with KW-200 alone reveals normal duodenum (D), jejunum (E) and ileum (F) with long and slender villi. (G-I) Calf inoculated with RVA displays severe villi atrophy and crypt hyperplasia in the duodenum (G), jejunum (H) and ileum (I). (J-L) Calf treated with KW-200 after induction of rotavirus diarrhea exhibits moderate restored villi and crypt in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and KW-200-treated groups. (A-C) Control calf shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Calf treated with KW-200 alone reveals normal duodenum (D), jejunum (E) and ileum (F) with long and slender villi. (G-I) Calf inoculated with RVA displays severe villi atrophy and crypt hyperplasia in the duodenum (G), jejunum (H) and ileum (I). (J-L) Calf treated with KW-200 after induction of rotavirus diarrhea exhibits moderate restored villi and crypt in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
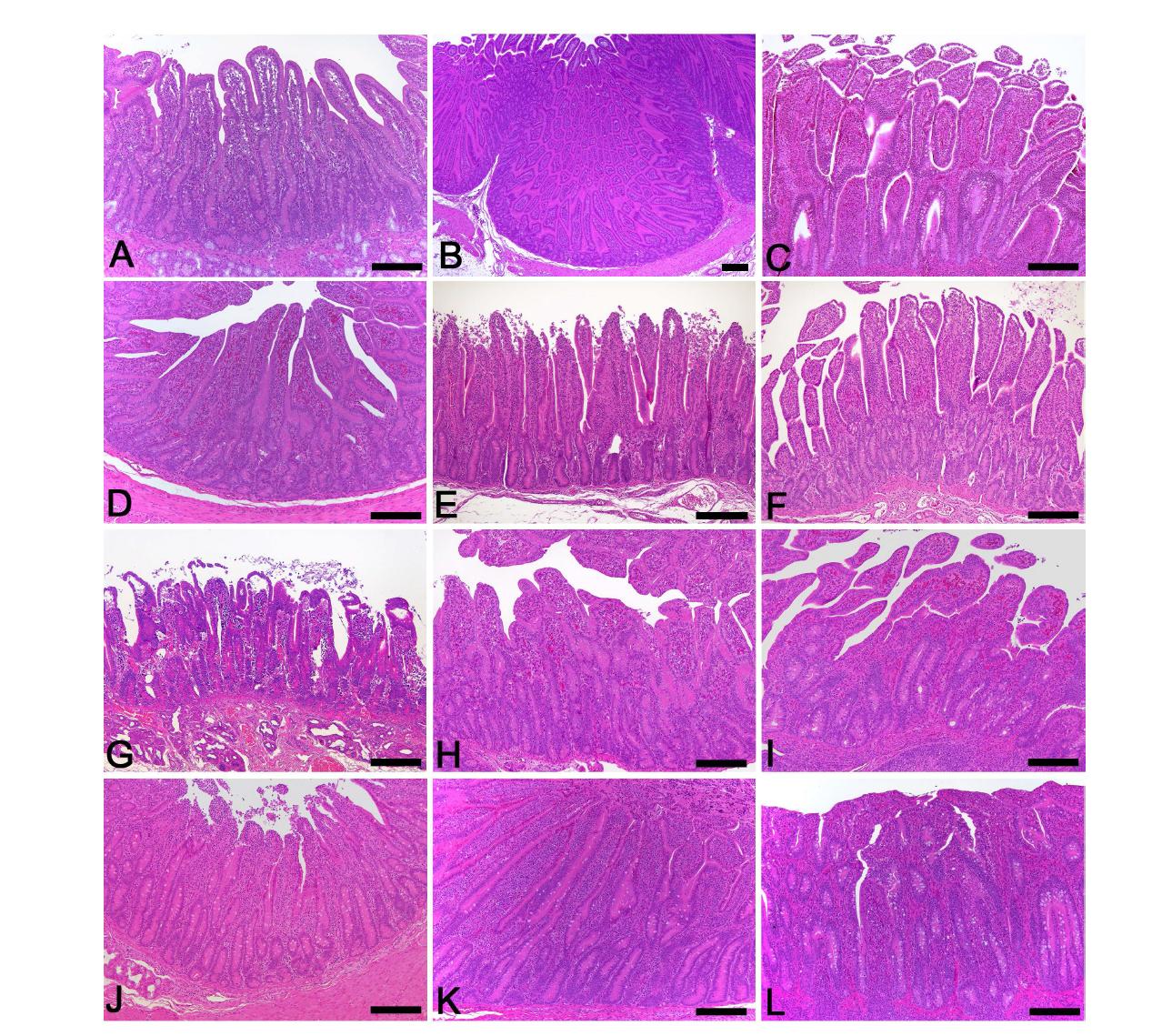 표
Histopathological changes of small intestine sampled from control and KW-200-treated groups. (A-C) Control calf shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Calf treated with KW-200 alone reveals normal duodenum (D), jejunum (E) and ileum (F) with long and slender villi. (G-I) Calf inoculated with bovine rotavirus and coronavirus displays severe villi atrophy and crypt hyperplasia in the duodenum (G), jejunum (H) and ileum (I). (J-L) Calf treated with KW-200 after diarrhea induction by experimental inoculation of both bovine rotavirus and coronavirus exhibits moderate restored villi and crypt in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and KW-200-treated groups. (A-C) Control calf shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Calf treated with KW-200 alone reveals normal duodenum (D), jejunum (E) and ileum (F) with long and slender villi. (G-I) Calf inoculated with bovine rotavirus and coronavirus displays severe villi atrophy and crypt hyperplasia in the duodenum (G), jejunum (H) and ileum (I). (J-L) Calf treated with KW-200 after diarrhea induction by experimental inoculation of both bovine rotavirus and coronavirus exhibits moderate restored villi and crypt in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
![Phylogenentic trees based on the full length nucleotide sequence of the 11 genomic segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains in comparison with reference RVA strains. The full length sequence of the 11 segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains were aligned, and phylogenetic trees were constructed using the neighbor-joining method with 1000 bootstrap replicates. Genetic distances were calculated using Kimura-2 correction parameter at the nucleotide level. Phylogenentic trees based on the full length nucleotide sequence of the 11 genomic segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains in comparison with reference RVA strains. The full length sequence of the 11 segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains were aligned, and phylogenetic trees were constructed using the neighbor-joining method with 1000 bootstrap replicates. Genetic distances were calculated using Kimura-2 correction parameter at the nucleotide level.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_200_image_156.jpg) 표
Phylogenentic trees based on the full length nucleotide sequence of the 11 genomic segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains in comparison with reference RVA strains. The full length sequence of the 11 segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains were aligned, and phylogenetic trees were constructed using the neighbor-joining method with 1000 bootstrap replicates. Genetic distances were calculated using Kimura-2 correction parameter at the nucleotide level.
표
Phylogenentic trees based on the full length nucleotide sequence of the 11 genomic segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains in comparison with reference RVA strains. The full length sequence of the 11 segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains were aligned, and phylogenetic trees were constructed using the neighbor-joining method with 1000 bootstrap replicates. Genetic distances were calculated using Kimura-2 correction parameter at the nucleotide level.
![Histopathological changes in the small intestine of calves and piglets infected with the bovine G5P[7] RVA strain K5. (A and B) Duodenum sampled from mock-inoculated piglet (A) and calf (B) had normal long slender villi (up-down arrow) and short crypt (up-wards arrow) in the mucosal membrane. (C) Duodenum sampled from a virus-infected piglet at DPI 3 showed severe villous atrophy (up-down arrow), villous fusion (arrows), and increased crypt depth (up-wards arrow). (D) Duodenum sampled from a virus-infected calf at DPI 3 had severe villous atrophy (up-down arrow), villous fusions (arrows) and increased crypt depth (up-wards arrow). Hematoxyline and eosin stain. Bars denote 400 μm. Histopathological changes in the small intestine of calves and piglets infected with the bovine G5P[7] RVA strain K5. (A and B) Duodenum sampled from mock-inoculated piglet (A) and calf (B) had normal long slender villi (up-down arrow) and short crypt (up-wards arrow) in the mucosal membrane. (C) Duodenum sampled from a virus-infected piglet at DPI 3 showed severe villous atrophy (up-down arrow), villous fusion (arrows), and increased crypt depth (up-wards arrow). (D) Duodenum sampled from a virus-infected calf at DPI 3 had severe villous atrophy (up-down arrow), villous fusions (arrows) and increased crypt depth (up-wards arrow). Hematoxyline and eosin stain. Bars denote 400 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_201_image_157.jpg) 표
Histopathological changes in the small intestine of calves and piglets infected with the bovine G5P[7] RVA strain K5. (A and B) Duodenum sampled from mock-inoculated piglet (A) and calf (B) had normal long slender villi (up-down arrow) and short crypt (up-wards arrow) in the mucosal membrane. (C) Duodenum sampled from a virus-infected piglet at DPI 3 showed severe villous atrophy (up-down arrow), villous fusion (arrows), and increased crypt depth (up-wards arrow). (D) Duodenum sampled from a virus-infected calf at DPI 3 had severe villous atrophy (up-down arrow), villous fusions (arrows) and increased crypt depth (up-wards arrow). Hematoxyline and eosin stain. Bars denote 400 μm.
표
Histopathological changes in the small intestine of calves and piglets infected with the bovine G5P[7] RVA strain K5. (A and B) Duodenum sampled from mock-inoculated piglet (A) and calf (B) had normal long slender villi (up-down arrow) and short crypt (up-wards arrow) in the mucosal membrane. (C) Duodenum sampled from a virus-infected piglet at DPI 3 showed severe villous atrophy (up-down arrow), villous fusion (arrows), and increased crypt depth (up-wards arrow). (D) Duodenum sampled from a virus-infected calf at DPI 3 had severe villous atrophy (up-down arrow), villous fusions (arrows) and increased crypt depth (up-wards arrow). Hematoxyline and eosin stain. Bars denote 400 μm.
![Distribution of RVA antigen-positive cells of porcine and bovine G5P[7] strains in the duodenum. (A and B) Duodenum sampled at DPI 3 from a piglet (A) and a calf (B) infected with bovine K5 strain had antigen-positive cells (arrows) in the villi. (C and D) K71 RVA antigen-positive cells (arrows) were detected in the villi of duodenum sampled from a virus inoculated piglet at DPI 1 (C), but not in those from a virus inoculated calf (D). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU. Bars denote 100 μm. Distribution of RVA antigen-positive cells of porcine and bovine G5P[7] strains in the duodenum. (A and B) Duodenum sampled at DPI 3 from a piglet (A) and a calf (B) infected with bovine K5 strain had antigen-positive cells (arrows) in the villi. (C and D) K71 RVA antigen-positive cells (arrows) were detected in the villi of duodenum sampled from a virus inoculated piglet at DPI 1 (C), but not in those from a virus inoculated calf (D). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU. Bars denote 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_207_image_163.jpg) 표
Distribution of RVA antigen-positive cells of porcine and bovine G5P[7] strains in the duodenum. (A and B) Duodenum sampled at DPI 3 from a piglet (A) and a calf (B) infected with bovine K5 strain had antigen-positive cells (arrows) in the villi. (C and D) K71 RVA antigen-positive cells (arrows) were detected in the villi of duodenum sampled from a virus inoculated piglet at DPI 1 (C), but not in those from a virus inoculated calf (D). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU. Bars denote 100 μm.
표
Distribution of RVA antigen-positive cells of porcine and bovine G5P[7] strains in the duodenum. (A and B) Duodenum sampled at DPI 3 from a piglet (A) and a calf (B) infected with bovine K5 strain had antigen-positive cells (arrows) in the villi. (C and D) K71 RVA antigen-positive cells (arrows) were detected in the villi of duodenum sampled from a virus inoculated piglet at DPI 1 (C), but not in those from a virus inoculated calf (D). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU. Bars denote 100 μm.
![Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with porcine G5P[7] RVA strain K71. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and RVA antigen-positive cells (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrows) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal against the VP6 protein of strain OSU (C, F, I and L). Bars denote 100 μm. Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with porcine G5P[7] RVA strain K71. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and RVA antigen-positive cells (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrows) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal against the VP6 protein of strain OSU (C, F, I and L). Bars denote 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_208_image_164.jpg) 표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with porcine G5P[7] RVA strain K71. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and RVA antigen-positive cells (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrows) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal against the VP6 protein of strain OSU (C, F, I and L). Bars denote 100 μm.
표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with porcine G5P[7] RVA strain K71. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and RVA antigen-positive cells (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrows) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal against the VP6 protein of strain OSU (C, F, I and L). Bars denote 100 μm.
![Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and an RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm. Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and an RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_209_image_165.jpg) 표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and an RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.
표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and an RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.
![Extra-intestinal histopathological changes and distribution of antigen-positive cells in calves infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated calf (A), MLN from a virus-inoculated calf showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated calf revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated calf showed interstitial pneumonia (arrows) (E) and RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal hepatocytes (arrows) from a mock-inoculated calf (G), liver sampled from a virus-inoculated calf showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated calf had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated calf displayed epithelial degeneration (arrowhead) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm. Extra-intestinal histopathological changes and distribution of antigen-positive cells in calves infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated calf (A), MLN from a virus-inoculated calf showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated calf revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated calf showed interstitial pneumonia (arrows) (E) and RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal hepatocytes (arrows) from a mock-inoculated calf (G), liver sampled from a virus-inoculated calf showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated calf had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated calf displayed epithelial degeneration (arrowhead) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_210_image_166.jpg) 표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in calves infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated calf (A), MLN from a virus-inoculated calf showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated calf revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated calf showed interstitial pneumonia (arrows) (E) and RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal hepatocytes (arrows) from a mock-inoculated calf (G), liver sampled from a virus-inoculated calf showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated calf had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated calf displayed epithelial degeneration (arrowhead) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.
표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in calves infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated calf (A), MLN from a virus-inoculated calf showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated calf revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated calf showed interstitial pneumonia (arrows) (E) and RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal hepatocytes (arrows) from a mock-inoculated calf (G), liver sampled from a virus-inoculated calf showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated calf had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated calf displayed epithelial degeneration (arrowhead) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.
![Quantification of RVA RNA copy numbers by SYBR Green real-time RT-PCR in the feces, serum, mesenteric lymph node (MLN), liver, lung, and choroid plexus. (A) The sequential changes of viral RNA copy numbers of a porcine G5P[7] strain in the feces, serum, MLN, liver, lung and choroid plexus sampled from virus-infected piglets. Fecal specimens sampled from porcine G5P[7] K71 strain-inoculated calves were also examined by real-time RT-PCR. Note that viral RNA copy numbers in the feces from virus-inoculated calves were markedly lower than those from virus-inoculated piglets. (B and C) The sequential changes of viral RNA copy numbers in the feces, serum, MLN, liver, lung, and choroid plexus sampled from virus-inoculated piglets (B) or calves (C) were similar to each other. The geometric means of virus RNA copy numbers per mg of each sample are displayed. Quantification of RVA RNA copy numbers by SYBR Green real-time RT-PCR in the feces, serum, mesenteric lymph node (MLN), liver, lung, and choroid plexus. (A) The sequential changes of viral RNA copy numbers of a porcine G5P[7] strain in the feces, serum, MLN, liver, lung and choroid plexus sampled from virus-infected piglets. Fecal specimens sampled from porcine G5P[7] K71 strain-inoculated calves were also examined by real-time RT-PCR. Note that viral RNA copy numbers in the feces from virus-inoculated calves were markedly lower than those from virus-inoculated piglets. (B and C) The sequential changes of viral RNA copy numbers in the feces, serum, MLN, liver, lung, and choroid plexus sampled from virus-inoculated piglets (B) or calves (C) were similar to each other. The geometric means of virus RNA copy numbers per mg of each sample are displayed.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_213_image_169.jpg) 표
Quantification of RVA RNA copy numbers by SYBR Green real-time RT-PCR in the feces, serum, mesenteric lymph node (MLN), liver, lung, and choroid plexus. (A) The sequential changes of viral RNA copy numbers of a porcine G5P[7] strain in the feces, serum, MLN, liver, lung and choroid plexus sampled from virus-infected piglets. Fecal specimens sampled from porcine G5P[7] K71 strain-inoculated calves were also examined by real-time RT-PCR. Note that viral RNA copy numbers in the feces from virus-inoculated calves were markedly lower than those from virus-inoculated piglets. (B and C) The sequential changes of viral RNA copy numbers in the feces, serum, MLN, liver, lung, and choroid plexus sampled from virus-inoculated piglets (B) or calves (C) were similar to each other. The geometric means of virus RNA copy numbers per mg of each sample are displayed.
표
Quantification of RVA RNA copy numbers by SYBR Green real-time RT-PCR in the feces, serum, mesenteric lymph node (MLN), liver, lung, and choroid plexus. (A) The sequential changes of viral RNA copy numbers of a porcine G5P[7] strain in the feces, serum, MLN, liver, lung and choroid plexus sampled from virus-infected piglets. Fecal specimens sampled from porcine G5P[7] K71 strain-inoculated calves were also examined by real-time RT-PCR. Note that viral RNA copy numbers in the feces from virus-inoculated calves were markedly lower than those from virus-inoculated piglets. (B and C) The sequential changes of viral RNA copy numbers in the feces, serum, MLN, liver, lung, and choroid plexus sampled from virus-inoculated piglets (B) or calves (C) were similar to each other. The geometric means of virus RNA copy numbers per mg of each sample are displayed.
 표
Duodenum, Jejunum, Ileum: A-C, Piglets inoculated with G5P7 rotavirus (5 x 104/ml virus titer). A Moderate changes in the duodenum. B Moderate changes in the jejunum. C Moderate changes in the ileum. D-F, G5P7 (5 x 10/ml5) strain inoculated piglets. D Severe villi atrophy and villi hyperplasia in the duodenum. E. Moderate villi atrophy and villi hyperplasia in the jejunum. F. Moderate villi atrophy and crypt hyperplasia in the ileum. G-I, G5P7(5x106/ml) inoculated piglets. G Severe villi atrophy and villi hyperplasia in the duodenum. H Severe villi atrophy and villi hyperplasia in the jejunum. I Severe villi atrophy and crypt hyperplasia in the ileum. J-L, G5P7(5x107/ml) inoculated piglets. J Severe villi atrophy and crypt hyperplasia in the duodenum. K Severe villi atrophy and crypt hyperplasia in the jejunum. L Marked villi atrophy and crypt hyperplasia in the ileum. Hematoxylin and eosin stain. Bar: A -L, 150㎛.
표
Duodenum, Jejunum, Ileum: A-C, Piglets inoculated with G5P7 rotavirus (5 x 104/ml virus titer). A Moderate changes in the duodenum. B Moderate changes in the jejunum. C Moderate changes in the ileum. D-F, G5P7 (5 x 10/ml5) strain inoculated piglets. D Severe villi atrophy and villi hyperplasia in the duodenum. E. Moderate villi atrophy and villi hyperplasia in the jejunum. F. Moderate villi atrophy and crypt hyperplasia in the ileum. G-I, G5P7(5x106/ml) inoculated piglets. G Severe villi atrophy and villi hyperplasia in the duodenum. H Severe villi atrophy and villi hyperplasia in the jejunum. I Severe villi atrophy and crypt hyperplasia in the ileum. J-L, G5P7(5x107/ml) inoculated piglets. J Severe villi atrophy and crypt hyperplasia in the duodenum. K Severe villi atrophy and crypt hyperplasia in the jejunum. L Marked villi atrophy and crypt hyperplasia in the ileum. Hematoxylin and eosin stain. Bar: A -L, 150㎛.
![Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G8P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy and crypt hyperplasia in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛. Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G8P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy and crypt hyperplasia in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_47_image_8.jpg) 표
Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G8P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy and crypt hyperplasia in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.
표
Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G8P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy and crypt hyperplasia in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.
![Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G5P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy ,crypt hyperplasia and lyphoid cell infiltratioin in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛. Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G5P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy ,crypt hyperplasia and lyphoid cell infiltratioin in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_48_image_9.jpg) 표
Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G5P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy ,crypt hyperplasia and lyphoid cell infiltratioin in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.
표
Duodenum, Jejunum and Ileum: calf. A -C, Mock-inoculated control calf. Normal duodenum (A ), jejunum (B) and ileum (C) showing long and slender villi. D-L, G5P[7] strain inoculated calves. D Mild villi atrophy and crypt hyperplasia in the duodenum at PID(Post-inoculation day) 1. E Mild villi atrophy and crypt hyperplasia in the jejunum at PID 1. F Mild villi atrophy and crypt hyperplasia in the ileum at PID 1. G Moderate villi atrophy ,crypt hyperplasia and lyphoid cell infiltratioin in the duodenum at PID 3. H Moderate villi atrophy and crypt hyperplasia in the jejunum at PID 3. I Moderate villi atrophy and crypt hyperplasia in the ileum at PID 3. J Severe villi atrophy and crypt hyperplasia in the duodenum at PID 7. K Severe villi atrophy and crypt hyperplasia in the jejunum at PID 7. L Severe villi atrophy and crypt hyperplasia in the ileum at PID 7. Hematoxylin and eosin stain. Bar: A -L, 200㎛.
![Antigen localization of rotaviruses; calf A-C Heterologous G5P[7] strain-inoculated calf. A -B. Lung sampled at PID(Post-inoculation day) 7 from calf inoculated with the heterolotous bovine rotavirus G5P[7] strain. C. Liver sampled at PID 7 from calf inoculated with the geterologous bovine rotavirus G5P[7]. A-B, 100㎛ C 200㎛. Antigen localization of rotaviruses; calf A-C Heterologous G5P[7] strain-inoculated calf. A -B. Lung sampled at PID(Post-inoculation day) 7 from calf inoculated with the heterolotous bovine rotavirus G5P[7] strain. C. Liver sampled at PID 7 from calf inoculated with the geterologous bovine rotavirus G5P[7]. A-B, 100㎛ C 200㎛.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_49_image_10.jpg) 표
Antigen localization of rotaviruses; calf A-C Heterologous G5P[7] strain-inoculated calf. A -B. Lung sampled at PID(Post-inoculation day) 7 from calf inoculated with the heterolotous bovine rotavirus G5P[7] strain. C. Liver sampled at PID 7 from calf inoculated with the geterologous bovine rotavirus G5P[7]. A-B, 100㎛ C 200㎛.
표
Antigen localization of rotaviruses; calf A-C Heterologous G5P[7] strain-inoculated calf. A -B. Lung sampled at PID(Post-inoculation day) 7 from calf inoculated with the heterolotous bovine rotavirus G5P[7] strain. C. Liver sampled at PID 7 from calf inoculated with the geterologous bovine rotavirus G5P[7]. A-B, 100㎛ C 200㎛.
![Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain. D. Duodenum showing severe villi atrophy and crypt cell hyperplasia. E. Severe villi atrophy and crypt hyperplasia in the jejunum. F. Ileum showing severe villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC after diarrhea induction by a G5P[7] strain showing slight villi atrophy and crypt hyperplasia. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. H&E stain. Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain. D. Duodenum showing severe villi atrophy and crypt cell hyperplasia. E. Severe villi atrophy and crypt hyperplasia in the jejunum. F. Ileum showing severe villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC after diarrhea induction by a G5P[7] strain showing slight villi atrophy and crypt hyperplasia. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. H&E stain.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_89_image_53.jpg) 표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain. D. Duodenum showing severe villi atrophy and crypt cell hyperplasia. E. Severe villi atrophy and crypt hyperplasia in the jejunum. F. Ileum showing severe villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC after diarrhea induction by a G5P[7] strain showing slight villi atrophy and crypt hyperplasia. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. H&E stain.
표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain. D. Duodenum showing severe villi atrophy and crypt cell hyperplasia. E. Severe villi atrophy and crypt hyperplasia in the jejunum. F. Ileum showing severe villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC after diarrhea induction by a G5P[7] strain showing slight villi atrophy and crypt hyperplasia. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC after diarrhea induction by a G5P[7] strain showing mild villi atrophy and crypt hyperplasia. H&E stain.
![Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 200 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. (M-O) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus. Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 200 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. (M-O) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_91_image_55.jpg) 표
Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 200 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. (M-O) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
표
Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 200 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. (M-O) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
![Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain showing severe villi atrophy and crypt hyperplasia. (D-F) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain exhibiting slight to mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC showing slight to mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC revealing slight to mild villi atrophy and crypt hyperplasia. Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain showing severe villi atrophy and crypt hyperplasia. (D-F) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain exhibiting slight to mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC showing slight to mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC revealing slight to mild villi atrophy and crypt hyperplasia.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_98_image_60.jpg) 표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain showing severe villi atrophy and crypt hyperplasia. (D-F) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain exhibiting slight to mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC showing slight to mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC revealing slight to mild villi atrophy and crypt hyperplasia.
표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain showing severe villi atrophy and crypt hyperplasia. (D-F) Duodenum, jejunum, and ileum sampled from piglets treated with 100 mg/ml JSC after diarrhea induction by a G5P[7] strain exhibiting slight to mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with 200 mg/ml JSC showing slight to mild villi atrophy and crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets treated with 400 mg/ml JSC revealing slight to mild villi atrophy and crypt hyperplasia.
![Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus. Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_100_image_62.jpg) 표
Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
표
Antigen distribution in the duodenum, jejunum, ileum. (A-C) Duodenum, jejunum and ileum sampled from mock-inoculated piglets showing negative reaction in the villi epithelium for rotavirus antigen. (D-E) Duodenum, jejunum and ileum sampled from piglets inoculated with a G5P[7] strain showing positive reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with 100 mg/ml JSC showing positive reactions in few of villi epithelium for rotavirus antigen. (J-K) Duodenum, jejunum and ileum sampled from piglets treated with 400 mg/ml JSC showing negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
![Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain exhibiting severe atrophy and fusion of villi. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in saline after diarrhea induction by the inoculation of a porcine rotavirus G5P[7] strain showing severe atrophy and fusion of villi, crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets starved for 1 day before treatment of ST-100 in saline revealing severe atrophy and fusion of villi. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in milk showing severe villi atrophy and fusion. H&E stain. Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain exhibiting severe atrophy and fusion of villi. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in saline after diarrhea induction by the inoculation of a porcine rotavirus G5P[7] strain showing severe atrophy and fusion of villi, crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets starved for 1 day before treatment of ST-100 in saline revealing severe atrophy and fusion of villi. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in milk showing severe villi atrophy and fusion. H&E stain.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_109_image_67.jpg) 표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain exhibiting severe atrophy and fusion of villi. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in saline after diarrhea induction by the inoculation of a porcine rotavirus G5P[7] strain showing severe atrophy and fusion of villi, crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets starved for 1 day before treatment of ST-100 in saline revealing severe atrophy and fusion of villi. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in milk showing severe villi atrophy and fusion. H&E stain.
표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. (A-C) Duodenum, jejunum, and ileum sampled from the normal piglets: Unaltered duodenum, jejunum & ileum showing long and slender villi. (D-F) Duodenum, jejunum, and ileum sampled from piglets inoculated with a G5P[7] strain exhibiting severe atrophy and fusion of villi. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in saline after diarrhea induction by the inoculation of a porcine rotavirus G5P[7] strain showing severe atrophy and fusion of villi, crypt hyperplasia. (J-L) Duodenum, jejunum, and ileum sampled from piglets starved for 1 day before treatment of ST-100 in saline revealing severe atrophy and fusion of villi. (M-O) Duodenum, jejunum, and ileum sampled from piglets treated with ST-100 in milk showing severe villi atrophy and fusion. H&E stain.
 표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. See normal and histopathological changes of small intestine in the upper figures. (A-C) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 100 mg/ml JSC and 3 g ST-100 showing mild villi atrophy and crypt hyperplasia. (E-F) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 200 mg/ml JSC and 3 g ST-100 exhibiting mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 400 mg/ml JSC and 3 g ST-100 revealing slight villi atrophy and crypt hyperplasia. H&E stain.
표
Comparison of hitological changes of small intestine sampled from normal piglets, and mock-treated and drug candidate-treated piglets after the induction of rotavirus diarrhea. See normal and histopathological changes of small intestine in the upper figures. (A-C) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 100 mg/ml JSC and 3 g ST-100 showing mild villi atrophy and crypt hyperplasia. (E-F) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 200 mg/ml JSC and 3 g ST-100 exhibiting mild villi atrophy and crypt hyperplasia. (G-I) Duodenum, jejunum, and ileum sampled from piglets treated with a mixture of 400 mg/ml JSC and 3 g ST-100 revealing slight villi atrophy and crypt hyperplasia. H&E stain.
 표
Antigen distribution in the duodenum, jejunum, ileum. See antigen appearance in the small intestine sampled from normal and virus-inoculated piglets, respectively, in the upper figures. (A-C) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 100 mg/ml JSC and 3 g ST-100 revealing negative reactions in the villi epithelium for rotavirus antigen. (D-F) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 200 mg/ml JSC and 3 g ST-100 exhibiting negative reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 400 mg/ml JSC and 3 g ST-100 having negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
표
Antigen distribution in the duodenum, jejunum, ileum. See antigen appearance in the small intestine sampled from normal and virus-inoculated piglets, respectively, in the upper figures. (A-C) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 100 mg/ml JSC and 3 g ST-100 revealing negative reactions in the villi epithelium for rotavirus antigen. (D-F) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 200 mg/ml JSC and 3 g ST-100 exhibiting negative reactions in the villi epithelium for rotavirus antigen. (G-I) Duodenum, jejunum and ileum sampled from piglets treated with a mixture of 400 mg/ml JSC and 3 g ST-100 having negative reactions in the villi epithelium for rotavirus antigen. Indirect immunofluorescence assay with monoclonal antibody specific to VP6 protein of group A rotavirus.
![Experimental design for evaluating anti-rotavirus effects of methanol-extracted KW-200. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg KW-200-treated, group IV was virus-inoculated and 200 mg KW-200-treated, and group V was virus-inoculated and 400 mg/mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for evaluating anti-rotavirus effects of methanol-extracted KW-200. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg KW-200-treated, group IV was virus-inoculated and 200 mg KW-200-treated, and group V was virus-inoculated and 400 mg/mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_121_image_78.jpg) 표
Experimental design for evaluating anti-rotavirus effects of methanol-extracted KW-200. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg KW-200-treated, group IV was virus-inoculated and 200 mg KW-200-treated, and group V was virus-inoculated and 400 mg/mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for evaluating anti-rotavirus effects of methanol-extracted KW-200. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg KW-200-treated, group IV was virus-inoculated and 200 mg KW-200-treated, and group V was virus-inoculated and 400 mg/mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
 표
Influence of KW-200 on rotavirus-induced expression of IL8, IL10, IFN-β, IFN-γ, and TNF-α in colostrums-deprived piglets. Comparison of mRNA expression levels of IL8, IL10, IFN-β, IFN-γ, and TNF-α with those of β-actin in each experimental group was determined by SYBR Green Real-time RT-PCR. Values are means ± S.D. (n=3) (*p< 0.05).
표
Influence of KW-200 on rotavirus-induced expression of IL8, IL10, IFN-β, IFN-γ, and TNF-α in colostrums-deprived piglets. Comparison of mRNA expression levels of IL8, IL10, IFN-β, IFN-γ, and TNF-α with those of β-actin in each experimental group was determined by SYBR Green Real-time RT-PCR. Values are means ± S.D. (n=3) (*p< 0.05).
![Experimental design for evaluating anti-rotavirus effects of KW-200 fraction. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 10 mg KW-200 fraction-treated, group IV was virus-inoculated and 25 mg KW-200 fraction-treated, and group V was virus-inoculated, 50 mg/mg KW-200 fraction-treated. KW-200 fraction-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for evaluating anti-rotavirus effects of KW-200 fraction. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 10 mg KW-200 fraction-treated, group IV was virus-inoculated and 25 mg KW-200 fraction-treated, and group V was virus-inoculated, 50 mg/mg KW-200 fraction-treated. KW-200 fraction-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_134_image_93.jpg) 표
Experimental design for evaluating anti-rotavirus effects of KW-200 fraction. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 10 mg KW-200 fraction-treated, group IV was virus-inoculated and 25 mg KW-200 fraction-treated, and group V was virus-inoculated, 50 mg/mg KW-200 fraction-treated. KW-200 fraction-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for evaluating anti-rotavirus effects of KW-200 fraction. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 10 mg KW-200 fraction-treated, group IV was virus-inoculated and 25 mg KW-200 fraction-treated, and group V was virus-inoculated, 50 mg/mg KW-200 fraction-treated. KW-200 fraction-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
 표
Histopathological changes of small intestine sampled from control and KW-200 fraction-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 10 mg/ml KW-200 fraction exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 25 mg/ml KW-200 fraction reveals slight improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). (M-O) Piglet treated with 50 mg/ml KW-200 shows moderate restored villi and crypt in the duodenum (M), jejunum (N) and ileum (O). Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and KW-200 fraction-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 10 mg/ml KW-200 fraction exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 25 mg/ml KW-200 fraction reveals slight improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). (M-O) Piglet treated with 50 mg/ml KW-200 shows moderate restored villi and crypt in the duodenum (M), jejunum (N) and ileum (O). Bars = 200 μm.
 표
Influence of KW-200 fraction on rotavirus-induced expression of TNF-α (A), IFN-β (B), NF-κB (C), and IL8 (D) in colostrums-deprived piglets. Comparison of mRNA expression levels of TNF-α (A), IFN-β (B), NF-κB (C), and IL8 (D) with those of β-actin in each experimental group was determined by SYBR Green Real-time RT-PCR.
표
Influence of KW-200 fraction on rotavirus-induced expression of TNF-α (A), IFN-β (B), NF-κB (C), and IL8 (D) in colostrums-deprived piglets. Comparison of mRNA expression levels of TNF-α (A), IFN-β (B), NF-κB (C), and IL8 (D) with those of β-actin in each experimental group was determined by SYBR Green Real-time RT-PCR.
![Experimental design for evaluating anti-rotavirus effects of glycyrrhizin. Experimental animals were randomly allocated into 4 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg glycyrrhizin-treated, and group IV was virus-inoculated and 200 mg glycyrrhizin-treated. Glycyrrhizin-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for evaluating anti-rotavirus effects of glycyrrhizin. Experimental animals were randomly allocated into 4 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg glycyrrhizin-treated, and group IV was virus-inoculated and 200 mg glycyrrhizin-treated. Glycyrrhizin-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_140_image_98.jpg) 표
Experimental design for evaluating anti-rotavirus effects of glycyrrhizin. Experimental animals were randomly allocated into 4 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg glycyrrhizin-treated, and group IV was virus-inoculated and 200 mg glycyrrhizin-treated. Glycyrrhizin-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for evaluating anti-rotavirus effects of glycyrrhizin. Experimental animals were randomly allocated into 4 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 100 mg glycyrrhizin-treated, and group IV was virus-inoculated and 200 mg glycyrrhizin-treated. Glycyrrhizin-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
 표
Histopathological changes of small intestine sampled from control and glycyrrhizin-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 100 mg/ml glycyrrhizin exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 200 mg/ml glycyrrhizin reveals no improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and glycyrrhizin-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 100 mg/ml glycyrrhizin exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 200 mg/ml glycyrrhizin reveals no improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
![Histopathological changes in the small intestine of piglets inoculated with PRG942 (G9P[23]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-inoculated piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm. Histopathological changes in the small intestine of piglets inoculated with PRG942 (G9P[23]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-inoculated piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_147_image_105.jpg) 표
Histopathological changes in the small intestine of piglets inoculated with PRG942 (G9P[23]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-inoculated piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.
표
Histopathological changes in the small intestine of piglets inoculated with PRG942 (G9P[23]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-inoculated piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.
![Histopathological changes in the small intestine of piglets inoculated with PRG9121 (G9P[7]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-infected piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet infected by PRG9121 (G9P[7]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm. Histopathological changes in the small intestine of piglets inoculated with PRG9121 (G9P[7]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-infected piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet infected by PRG9121 (G9P[7]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_148_image_106.jpg) 표
Histopathological changes in the small intestine of piglets inoculated with PRG9121 (G9P[7]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-infected piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet infected by PRG9121 (G9P[7]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.
표
Histopathological changes in the small intestine of piglets inoculated with PRG9121 (G9P[7]) strain. Duodenum (A), jejunum (B), and ileum (C) from a mock-infected piglet had normal structure of mucosal membrane. Duodenum (D), jejunum (E), and ileum (F) sampled from a piglet infected by PRG9121 (G9P[7]) strain at DPI 1 showed mild to moderate mucosal changes. Duodenum (G), jejunum (H), and ileum (I) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked mucosal changes including the widespread villous atrophy (up-down arrow) and fusion (arrows), and increased crypt depth (up-wards arrow). Duodenum (J), jejunum (K), and ileum (L) sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 7 had marked mucosal changes. Samples were stained with hematoxylin and eosin stain. Bars A. L = 200 μm.
![Distribution of RVA-positive cells of PRG942 (G9P[23]) and PRG9121 (G9P[7]) strains in the duodenum. Piglets inoculated with either chloroform-inactivated PRG942 (G9P[23]) strain (A) or PRG9121 (G9P[7]) strain (B) contained no RVA-positive cells in the villi of each duodenum. RVA-positive cells (arrows) were scattered in the villi of duodenum sampled from piglets inoculated with PRG942 (G9P[23]) strain (C) or PRG9121 (G9P[7]) strain (D) at DPI 3. Indirect immunofluorescence assay was performed with monoclonal antibody against the VP6 protein of strain OSU. Bars A. D = 100 μm. Distribution of RVA-positive cells of PRG942 (G9P[23]) and PRG9121 (G9P[7]) strains in the duodenum. Piglets inoculated with either chloroform-inactivated PRG942 (G9P[23]) strain (A) or PRG9121 (G9P[7]) strain (B) contained no RVA-positive cells in the villi of each duodenum. RVA-positive cells (arrows) were scattered in the villi of duodenum sampled from piglets inoculated with PRG942 (G9P[23]) strain (C) or PRG9121 (G9P[7]) strain (D) at DPI 3. Indirect immunofluorescence assay was performed with monoclonal antibody against the VP6 protein of strain OSU. Bars A. D = 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_149_image_107.jpg) 표
Distribution of RVA-positive cells of PRG942 (G9P[23]) and PRG9121 (G9P[7]) strains in the duodenum. Piglets inoculated with either chloroform-inactivated PRG942 (G9P[23]) strain (A) or PRG9121 (G9P[7]) strain (B) contained no RVA-positive cells in the villi of each duodenum. RVA-positive cells (arrows) were scattered in the villi of duodenum sampled from piglets inoculated with PRG942 (G9P[23]) strain (C) or PRG9121 (G9P[7]) strain (D) at DPI 3. Indirect immunofluorescence assay was performed with monoclonal antibody against the VP6 protein of strain OSU. Bars A. D = 100 μm.
표
Distribution of RVA-positive cells of PRG942 (G9P[23]) and PRG9121 (G9P[7]) strains in the duodenum. Piglets inoculated with either chloroform-inactivated PRG942 (G9P[23]) strain (A) or PRG9121 (G9P[7]) strain (B) contained no RVA-positive cells in the villi of each duodenum. RVA-positive cells (arrows) were scattered in the villi of duodenum sampled from piglets inoculated with PRG942 (G9P[23]) strain (C) or PRG9121 (G9P[7]) strain (D) at DPI 3. Indirect immunofluorescence assay was performed with monoclonal antibody against the VP6 protein of strain OSU. Bars A. D = 100 μm.
![Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG942 (G9P[23]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (D) Normal lung tissue sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissue sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigens were detected in a pneumocyte (arrowhead) and lymphoid cell (arrow) in the lung from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrow). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a hepatocyte (arrow) in the liver from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigens were detected in the epithelial cells (arrow) of the choroid plexus from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. Bars A- L = 100 μm. Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG942 (G9P[23]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (D) Normal lung tissue sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissue sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigens were detected in a pneumocyte (arrowhead) and lymphoid cell (arrow) in the lung from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrow). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a hepatocyte (arrow) in the liver from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigens were detected in the epithelial cells (arrow) of the choroid plexus from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. Bars A- L = 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_153_image_111.jpg) 표
Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG942 (G9P[23]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (D) Normal lung tissue sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissue sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigens were detected in a pneumocyte (arrowhead) and lymphoid cell (arrow) in the lung from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrow). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a hepatocyte (arrow) in the liver from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigens were detected in the epithelial cells (arrow) of the choroid plexus from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. Bars A- L = 100 μm.
표
Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG942 (G9P[23]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (D) Normal lung tissue sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissue sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigens were detected in a pneumocyte (arrowhead) and lymphoid cell (arrow) in the lung from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrow). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a hepatocyte (arrow) in the liver from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigens were detected in the epithelial cells (arrow) of the choroid plexus from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3. Bars A- L = 100 μm.
![Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG9121 (G9P[7]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (D) Normal lung tissues sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissues sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigen was detected in a pneumocyte (arrow) in the lung from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrows). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a few hepatocytes (arrows) in the liver from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigen was detected in a lymphoid cell (arrow) in the choroid plexus from a piglet inoculated with PRG9121 (G9P[7]) strain. Bars A- L = 100 μm. Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG9121 (G9P[7]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (D) Normal lung tissues sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissues sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigen was detected in a pneumocyte (arrow) in the lung from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrows). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a few hepatocytes (arrows) in the liver from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigen was detected in a lymphoid cell (arrow) in the choroid plexus from a piglet inoculated with PRG9121 (G9P[7]) strain. Bars A- L = 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_154_image_112.jpg) 표
Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG9121 (G9P[7]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (D) Normal lung tissues sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissues sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigen was detected in a pneumocyte (arrow) in the lung from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrows). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a few hepatocytes (arrows) in the liver from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigen was detected in a lymphoid cell (arrow) in the choroid plexus from a piglet inoculated with PRG9121 (G9P[7]) strain. Bars A- L = 100 μm.
표
Histopathological findings and antigen distribution in the extra-intestinal organs of piglets inoculated with PRG9121 (G9P[7]) strain. (A) Normal mesenteric lymph node (MLN) sampled from a mock-inoculated piglet showed intact cortex with densely concentrated lymphocytes (asterisk). (B) MLN sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed marked lymphoid cell depletion in the cortex (asterisk). (C) RVA-positive lymphoid cells (arrows) were scattered in the MLN from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (D) Normal lung tissues sampled from a mock-inoculated piglet had thin alveolar walls (arrows). (E) Lung tissues sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed interstitial pneumonia (arrows). (F) RVA antigen was detected in a pneumocyte (arrow) in the lung from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (G) Normal liver sampled from a mock-inoculated piglet showed fat-stored hepatocytes (arrows). (H) Multiple necrotic hepatocytes scattered in the liver sampled from a piglet inoculated with PRG942 (G9P[23]) strain at DPI 3 showed pyknosis of nucleus and increased eosinophilia of cytoplasm (arrows). (I) RVA antigens were detected in a few hepatocytes (arrows) in the liver from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3. (J) Intact epithelium (arrowheads) was observed in the choroid plexus sampled from a mock-inoculated piglet. (K) Choroid plexus sampled from a piglet inoculated with PRG9121 (G9P[7]) strain at DPI 3 showed epithelial degeneration (arrowheads), necrosis (arrows), and lymphocyte infiltration (double arrows). (L) RVA antigen was detected in a lymphoid cell (arrow) in the choroid plexus from a piglet inoculated with PRG9121 (G9P[7]) strain. Bars A- L = 100 μm.
![Quantification of RVA RNA by SYBR Green real-time RT-PCR. Real-time RT-PCR was performed with the feces, serum, mesenteric lymph node (MLN), liver, lung, choroid plexus, and nasal swab sampled from piglets inoculated with porcine G9 RVA strain PRG942 (G9P[23]) (A) and PRG9121 (G9P[7]) (B). The geometric means of viral RNA copy number per mg of tissue are displayed. Quantification of RVA RNA by SYBR Green real-time RT-PCR. Real-time RT-PCR was performed with the feces, serum, mesenteric lymph node (MLN), liver, lung, choroid plexus, and nasal swab sampled from piglets inoculated with porcine G9 RVA strain PRG942 (G9P[23]) (A) and PRG9121 (G9P[7]) (B). The geometric means of viral RNA copy number per mg of tissue are displayed.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_155_image_113.jpg) 표
Quantification of RVA RNA by SYBR Green real-time RT-PCR. Real-time RT-PCR was performed with the feces, serum, mesenteric lymph node (MLN), liver, lung, choroid plexus, and nasal swab sampled from piglets inoculated with porcine G9 RVA strain PRG942 (G9P[23]) (A) and PRG9121 (G9P[7]) (B). The geometric means of viral RNA copy number per mg of tissue are displayed.
표
Quantification of RVA RNA by SYBR Green real-time RT-PCR. Real-time RT-PCR was performed with the feces, serum, mesenteric lymph node (MLN), liver, lung, choroid plexus, and nasal swab sampled from piglets inoculated with porcine G9 RVA strain PRG942 (G9P[23]) (A) and PRG9121 (G9P[7]) (B). The geometric means of viral RNA copy number per mg of tissue are displayed.
![Experimental design for evaluating anti-rotavirus effects of compression-extracted KW-200 immediately after harvesting. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 200 mg KW-200-treated, group IV was virus-inoculated and 400 mg KW-200-treated, gorup V was virus-inoculated and 600 mg KW-200-treated, and group VI was virus-inoculated and 800 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for evaluating anti-rotavirus effects of compression-extracted KW-200 immediately after harvesting. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 200 mg KW-200-treated, group IV was virus-inoculated and 400 mg KW-200-treated, gorup V was virus-inoculated and 600 mg KW-200-treated, and group VI was virus-inoculated and 800 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_156_image_114.jpg) 표
Experimental design for evaluating anti-rotavirus effects of compression-extracted KW-200 immediately after harvesting. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 200 mg KW-200-treated, group IV was virus-inoculated and 400 mg KW-200-treated, gorup V was virus-inoculated and 600 mg KW-200-treated, and group VI was virus-inoculated and 800 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for evaluating anti-rotavirus effects of compression-extracted KW-200 immediately after harvesting. Experimental animals were randomly allocated into 6 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 200 mg KW-200-treated, group IV was virus-inoculated and 400 mg KW-200-treated, gorup V was virus-inoculated and 600 mg KW-200-treated, and group VI was virus-inoculated and 800 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
![Experimental design for reevaluating anti-rotavirus effects on the various mixtures of ST-100 and JSC. This study was performed to address the points raised by reviewers. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for reevaluating anti-rotavirus effects on the various mixtures of ST-100 and JSC. This study was performed to address the points raised by reviewers. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_169_image_127.jpg) 표
Experimental design for reevaluating anti-rotavirus effects on the various mixtures of ST-100 and JSC. This study was performed to address the points raised by reviewers. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for reevaluating anti-rotavirus effects on the various mixtures of ST-100 and JSC. This study was performed to address the points raised by reviewers. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
 표
Histopathological changes of small intestine sampled from control and ST-100-JSC combination-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 12 g ST-100 exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 1600 mg JSC reveals moderate improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). (M-O) Piglet treated with 1600 mg JSC plus 12 g ST-100 shows markedly restored villi and crypt in the duodenum (M), jejunum (N) and ileum (O). (P-R) Piglet treated with 400 mg/ ml KW-200 shows unaltered duodenum (P), jejunum (Q) and ileum (R) with long and slender villi. Hematoxyline and eosin stain. Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and ST-100-JSC combination-treated groups. (A-C) Control piglet shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Piglet inoculated with RVA displays severe villi atropy and crypt hyperplasia in the duodenum (D), jejunum (E) and ileum (F). (G-I) Piglet treated with 12 g ST-100 exhibits no improvement of lesion changes in the duodenum (G), jejunum (H) and ileum (I). (J-L) Piglet treated with 1600 mg JSC reveals moderate improvement of lesion changes in the duodenum (J), jejunum (K) and ileum (L). (M-O) Piglet treated with 1600 mg JSC plus 12 g ST-100 shows markedly restored villi and crypt in the duodenum (M), jejunum (N) and ileum (O). (P-R) Piglet treated with 400 mg/ ml KW-200 shows unaltered duodenum (P), jejunum (Q) and ileum (R) with long and slender villi. Hematoxyline and eosin stain. Bars = 200 μm.
![Experimental design for reevaluating anti-rotavirus effects of KW-200 for addressing the points raised by reviewers. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 400 mg KW-200-treated, group IV was virus-inoculated and 800 mg KW-200-treated, and gorup V was virus-inoculated and 1600 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations. Experimental design for reevaluating anti-rotavirus effects of KW-200 for addressing the points raised by reviewers. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 400 mg KW-200-treated, group IV was virus-inoculated and 800 mg KW-200-treated, and gorup V was virus-inoculated and 1600 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_180_image_139.jpg) 표
Experimental design for reevaluating anti-rotavirus effects of KW-200 for addressing the points raised by reviewers. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 400 mg KW-200-treated, group IV was virus-inoculated and 800 mg KW-200-treated, and gorup V was virus-inoculated and 1600 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
표
Experimental design for reevaluating anti-rotavirus effects of KW-200 for addressing the points raised by reviewers. Experimental animals were randomly allocated into 5 groups in which group I was mock-inoculated and mock-treated, group II was virus-inoculated and mock-treated, group III was virus-inoculated and 400 mg KW-200-treated, group IV was virus-inoculated and 800 mg KW-200-treated, and gorup V was virus-inoculated and 1600 mg KW-200-treated. KW-200-treated groups were administrated for 7 days after occurrence of rotavirus diarrhea. All piglets inoculated with rotavirus G5P[7] strain K85 showed diarrhea at day post-inoculation (DPI) 2. Experiments were terminated at DPI 10. All animals died during the experimental period or at the termination of experiment were immediately necropsied and all specimens including the fecal samples, organs or tissues were sampled and used for further examinations.
 표
Experimental design for establishing human rotavirus experimental model. Piglets produced from sows by hysterectomy were kept in germ-free isolators. Experimental animals were inoculated with a human rotavirus strain Wa at 4 days old and euthanasied at day post-inoculation 1-6. Clinical signs including the diarrhea were monitored everyday and fecal specimens were sampled daily. All experimental animals. At necropsy, duodenum, jejunum and ileum were sampled for histopathological examination.
표
Experimental design for establishing human rotavirus experimental model. Piglets produced from sows by hysterectomy were kept in germ-free isolators. Experimental animals were inoculated with a human rotavirus strain Wa at 4 days old and euthanasied at day post-inoculation 1-6. Clinical signs including the diarrhea were monitored everyday and fecal specimens were sampled daily. All experimental animals. At necropsy, duodenum, jejunum and ileum were sampled for histopathological examination.
 표
Histopathological changes of small intestine sampled from control and KW-200-treated groups. (A-C) Control calf shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Calf treated with KW-200 alone reveals normal duodenum (D), jejunum (E) and ileum (F) with long and slender villi. (G-I) Calf inoculated with RVA displays severe villi atrophy and crypt hyperplasia in the duodenum (G), jejunum (H) and ileum (I). (J-L) Calf treated with KW-200 after induction of rotavirus diarrhea exhibits moderate restored villi and crypt in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and KW-200-treated groups. (A-C) Control calf shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Calf treated with KW-200 alone reveals normal duodenum (D), jejunum (E) and ileum (F) with long and slender villi. (G-I) Calf inoculated with RVA displays severe villi atrophy and crypt hyperplasia in the duodenum (G), jejunum (H) and ileum (I). (J-L) Calf treated with KW-200 after induction of rotavirus diarrhea exhibits moderate restored villi and crypt in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
 표
Histopathological changes of small intestine sampled from control and KW-200-treated groups. (A-C) Control calf shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Calf treated with KW-200 alone reveals normal duodenum (D), jejunum (E) and ileum (F) with long and slender villi. (G-I) Calf inoculated with bovine rotavirus and coronavirus displays severe villi atrophy and crypt hyperplasia in the duodenum (G), jejunum (H) and ileum (I). (J-L) Calf treated with KW-200 after diarrhea induction by experimental inoculation of both bovine rotavirus and coronavirus exhibits moderate restored villi and crypt in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
표
Histopathological changes of small intestine sampled from control and KW-200-treated groups. (A-C) Control calf shows unaltered duodenum (A), jejunum (B) and ileum (C) with long and slender villi. (D-F) Calf treated with KW-200 alone reveals normal duodenum (D), jejunum (E) and ileum (F) with long and slender villi. (G-I) Calf inoculated with bovine rotavirus and coronavirus displays severe villi atrophy and crypt hyperplasia in the duodenum (G), jejunum (H) and ileum (I). (J-L) Calf treated with KW-200 after diarrhea induction by experimental inoculation of both bovine rotavirus and coronavirus exhibits moderate restored villi and crypt in the duodenum (J), jejunum (K) and ileum (L). Bars = 200 μm.
![Phylogenentic trees based on the full length nucleotide sequence of the 11 genomic segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains in comparison with reference RVA strains. The full length sequence of the 11 segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains were aligned, and phylogenetic trees were constructed using the neighbor-joining method with 1000 bootstrap replicates. Genetic distances were calculated using Kimura-2 correction parameter at the nucleotide level. Phylogenentic trees based on the full length nucleotide sequence of the 11 genomic segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains in comparison with reference RVA strains. The full length sequence of the 11 segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains were aligned, and phylogenetic trees were constructed using the neighbor-joining method with 1000 bootstrap replicates. Genetic distances were calculated using Kimura-2 correction parameter at the nucleotide level.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_200_image_156.jpg) 표
Phylogenentic trees based on the full length nucleotide sequence of the 11 genomic segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains in comparison with reference RVA strains. The full length sequence of the 11 segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains were aligned, and phylogenetic trees were constructed using the neighbor-joining method with 1000 bootstrap replicates. Genetic distances were calculated using Kimura-2 correction parameter at the nucleotide level.
표
Phylogenentic trees based on the full length nucleotide sequence of the 11 genomic segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains in comparison with reference RVA strains. The full length sequence of the 11 segments of porcine G5P[7] K71 and porcine-like bovine G5P[7] K5 strains were aligned, and phylogenetic trees were constructed using the neighbor-joining method with 1000 bootstrap replicates. Genetic distances were calculated using Kimura-2 correction parameter at the nucleotide level.
![Histopathological changes in the small intestine of calves and piglets infected with the bovine G5P[7] RVA strain K5. (A and B) Duodenum sampled from mock-inoculated piglet (A) and calf (B) had normal long slender villi (up-down arrow) and short crypt (up-wards arrow) in the mucosal membrane. (C) Duodenum sampled from a virus-infected piglet at DPI 3 showed severe villous atrophy (up-down arrow), villous fusion (arrows), and increased crypt depth (up-wards arrow). (D) Duodenum sampled from a virus-infected calf at DPI 3 had severe villous atrophy (up-down arrow), villous fusions (arrows) and increased crypt depth (up-wards arrow). Hematoxyline and eosin stain. Bars denote 400 μm. Histopathological changes in the small intestine of calves and piglets infected with the bovine G5P[7] RVA strain K5. (A and B) Duodenum sampled from mock-inoculated piglet (A) and calf (B) had normal long slender villi (up-down arrow) and short crypt (up-wards arrow) in the mucosal membrane. (C) Duodenum sampled from a virus-infected piglet at DPI 3 showed severe villous atrophy (up-down arrow), villous fusion (arrows), and increased crypt depth (up-wards arrow). (D) Duodenum sampled from a virus-infected calf at DPI 3 had severe villous atrophy (up-down arrow), villous fusions (arrows) and increased crypt depth (up-wards arrow). Hematoxyline and eosin stain. Bars denote 400 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_201_image_157.jpg) 표
Histopathological changes in the small intestine of calves and piglets infected with the bovine G5P[7] RVA strain K5. (A and B) Duodenum sampled from mock-inoculated piglet (A) and calf (B) had normal long slender villi (up-down arrow) and short crypt (up-wards arrow) in the mucosal membrane. (C) Duodenum sampled from a virus-infected piglet at DPI 3 showed severe villous atrophy (up-down arrow), villous fusion (arrows), and increased crypt depth (up-wards arrow). (D) Duodenum sampled from a virus-infected calf at DPI 3 had severe villous atrophy (up-down arrow), villous fusions (arrows) and increased crypt depth (up-wards arrow). Hematoxyline and eosin stain. Bars denote 400 μm.
표
Histopathological changes in the small intestine of calves and piglets infected with the bovine G5P[7] RVA strain K5. (A and B) Duodenum sampled from mock-inoculated piglet (A) and calf (B) had normal long slender villi (up-down arrow) and short crypt (up-wards arrow) in the mucosal membrane. (C) Duodenum sampled from a virus-infected piglet at DPI 3 showed severe villous atrophy (up-down arrow), villous fusion (arrows), and increased crypt depth (up-wards arrow). (D) Duodenum sampled from a virus-infected calf at DPI 3 had severe villous atrophy (up-down arrow), villous fusions (arrows) and increased crypt depth (up-wards arrow). Hematoxyline and eosin stain. Bars denote 400 μm.
![Distribution of RVA antigen-positive cells of porcine and bovine G5P[7] strains in the duodenum. (A and B) Duodenum sampled at DPI 3 from a piglet (A) and a calf (B) infected with bovine K5 strain had antigen-positive cells (arrows) in the villi. (C and D) K71 RVA antigen-positive cells (arrows) were detected in the villi of duodenum sampled from a virus inoculated piglet at DPI 1 (C), but not in those from a virus inoculated calf (D). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU. Bars denote 100 μm. Distribution of RVA antigen-positive cells of porcine and bovine G5P[7] strains in the duodenum. (A and B) Duodenum sampled at DPI 3 from a piglet (A) and a calf (B) infected with bovine K5 strain had antigen-positive cells (arrows) in the villi. (C and D) K71 RVA antigen-positive cells (arrows) were detected in the villi of duodenum sampled from a virus inoculated piglet at DPI 1 (C), but not in those from a virus inoculated calf (D). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU. Bars denote 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_207_image_163.jpg) 표
Distribution of RVA antigen-positive cells of porcine and bovine G5P[7] strains in the duodenum. (A and B) Duodenum sampled at DPI 3 from a piglet (A) and a calf (B) infected with bovine K5 strain had antigen-positive cells (arrows) in the villi. (C and D) K71 RVA antigen-positive cells (arrows) were detected in the villi of duodenum sampled from a virus inoculated piglet at DPI 1 (C), but not in those from a virus inoculated calf (D). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU. Bars denote 100 μm.
표
Distribution of RVA antigen-positive cells of porcine and bovine G5P[7] strains in the duodenum. (A and B) Duodenum sampled at DPI 3 from a piglet (A) and a calf (B) infected with bovine K5 strain had antigen-positive cells (arrows) in the villi. (C and D) K71 RVA antigen-positive cells (arrows) were detected in the villi of duodenum sampled from a virus inoculated piglet at DPI 1 (C), but not in those from a virus inoculated calf (D). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU. Bars denote 100 μm.
![Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with porcine G5P[7] RVA strain K71. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and RVA antigen-positive cells (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrows) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal against the VP6 protein of strain OSU (C, F, I and L). Bars denote 100 μm. Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with porcine G5P[7] RVA strain K71. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and RVA antigen-positive cells (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrows) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal against the VP6 protein of strain OSU (C, F, I and L). Bars denote 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_208_image_164.jpg) 표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with porcine G5P[7] RVA strain K71. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and RVA antigen-positive cells (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrows) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal against the VP6 protein of strain OSU (C, F, I and L). Bars denote 100 μm.
표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with porcine G5P[7] RVA strain K71. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and RVA antigen-positive cells (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrows) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal against the VP6 protein of strain OSU (C, F, I and L). Bars denote 100 μm.
![Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and an RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm. Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and an RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_209_image_165.jpg) 표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and an RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.
표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in piglets infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated piglet (A), MLN from a virus-inoculated piglet showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated piglet revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated piglet showed interstitial pneumonia (arrows) (E) and an RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal fat-storing hepatocytes (arrows) from a mock-inoculated piglet (G), liver sampled from a virus-inoculated piglet showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated piglet had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated piglet displayed epithelial degeneration (arrowheads) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.
![Extra-intestinal histopathological changes and distribution of antigen-positive cells in calves infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated calf (A), MLN from a virus-inoculated calf showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated calf revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated calf showed interstitial pneumonia (arrows) (E) and RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal hepatocytes (arrows) from a mock-inoculated calf (G), liver sampled from a virus-inoculated calf showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated calf had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated calf displayed epithelial degeneration (arrowhead) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm. Extra-intestinal histopathological changes and distribution of antigen-positive cells in calves infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated calf (A), MLN from a virus-inoculated calf showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated calf revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated calf showed interstitial pneumonia (arrows) (E) and RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal hepatocytes (arrows) from a mock-inoculated calf (G), liver sampled from a virus-inoculated calf showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated calf had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated calf displayed epithelial degeneration (arrowhead) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_210_image_166.jpg) 표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in calves infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated calf (A), MLN from a virus-inoculated calf showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated calf revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated calf showed interstitial pneumonia (arrows) (E) and RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal hepatocytes (arrows) from a mock-inoculated calf (G), liver sampled from a virus-inoculated calf showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated calf had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated calf displayed epithelial degeneration (arrowhead) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.
표
Extra-intestinal histopathological changes and distribution of antigen-positive cells in calves infected with bovine G5P[7] RVA strain K5. (A-C) Compared to densely packed lymphocytes (asterisk) in the cortex of normal mesenteric lymph node (MLN) from a mock-inoculated calf (A), MLN from a virus-inoculated calf showed lymphoid cell depletion (asterisk) in the cortex (B) and RVA antigen-positive cells (arrows) (C). (D-F) Lung sampled from a mock-inoculated calf revealed normal thin alveolar wall (arrows) (D), whereas lung sampled from a virus-inoculated calf showed interstitial pneumonia (arrows) (E) and RVA antigen-positive reaction in the pneumocyte (arrowhead) and lymphoid cell (arrow) (F). (G-I) Compared to normal hepatocytes (arrows) from a mock-inoculated calf (G), liver sampled from a virus-inoculated calf showed multiple scattered necrotic hepatocytes (arrows) (H) and RVA antigen-positive cells (arrows) (I). (J-L) Choroid plexus sampled from a mock-inoculated calf had intact epithelium (arrowheads) (J), whereas choroid plexus sampled from a virus-inoculated calf displayed epithelial degeneration (arrowhead) and necrosis (arrows), and lymphoid cell infiltration (double arrow) into the tela choroidea (H), and RVA antigen-positive cells (arrow) (L). Hematoxylin and eosin stain (A, B, D, E, G, H, J, and K). Indirect immunofluorescence assay with monoclonal antibody against the VP6 protein of strain OSU (C, F, I, and L). Bars denote 100 μm.
![Quantification of RVA RNA copy numbers by SYBR Green real-time RT-PCR in the feces, serum, mesenteric lymph node (MLN), liver, lung, and choroid plexus. (A) The sequential changes of viral RNA copy numbers of a porcine G5P[7] strain in the feces, serum, MLN, liver, lung and choroid plexus sampled from virus-infected piglets. Fecal specimens sampled from porcine G5P[7] K71 strain-inoculated calves were also examined by real-time RT-PCR. Note that viral RNA copy numbers in the feces from virus-inoculated calves were markedly lower than those from virus-inoculated piglets. (B and C) The sequential changes of viral RNA copy numbers in the feces, serum, MLN, liver, lung, and choroid plexus sampled from virus-inoculated piglets (B) or calves (C) were similar to each other. The geometric means of virus RNA copy numbers per mg of each sample are displayed. Quantification of RVA RNA copy numbers by SYBR Green real-time RT-PCR in the feces, serum, mesenteric lymph node (MLN), liver, lung, and choroid plexus. (A) The sequential changes of viral RNA copy numbers of a porcine G5P[7] strain in the feces, serum, MLN, liver, lung and choroid plexus sampled from virus-infected piglets. Fecal specimens sampled from porcine G5P[7] K71 strain-inoculated calves were also examined by real-time RT-PCR. Note that viral RNA copy numbers in the feces from virus-inoculated calves were markedly lower than those from virus-inoculated piglets. (B and C) The sequential changes of viral RNA copy numbers in the feces, serum, MLN, liver, lung, and choroid plexus sampled from virus-inoculated piglets (B) or calves (C) were similar to each other. The geometric means of virus RNA copy numbers per mg of each sample are displayed.](https://nrms.kisti.re.kr/bitextimages/TRKO201500002491/TRKO201500002491_213_image_169.jpg) 표
Quantification of RVA RNA copy numbers by SYBR Green real-time RT-PCR in the feces, serum, mesenteric lymph node (MLN), liver, lung, and choroid plexus. (A) The sequential changes of viral RNA copy numbers of a porcine G5P[7] strain in the feces, serum, MLN, liver, lung and choroid plexus sampled from virus-infected piglets. Fecal specimens sampled from porcine G5P[7] K71 strain-inoculated calves were also examined by real-time RT-PCR. Note that viral RNA copy numbers in the feces from virus-inoculated calves were markedly lower than those from virus-inoculated piglets. (B and C) The sequential changes of viral RNA copy numbers in the feces, serum, MLN, liver, lung, and choroid plexus sampled from virus-inoculated piglets (B) or calves (C) were similar to each other. The geometric means of virus RNA copy numbers per mg of each sample are displayed.
표
Quantification of RVA RNA copy numbers by SYBR Green real-time RT-PCR in the feces, serum, mesenteric lymph node (MLN), liver, lung, and choroid plexus. (A) The sequential changes of viral RNA copy numbers of a porcine G5P[7] strain in the feces, serum, MLN, liver, lung and choroid plexus sampled from virus-infected piglets. Fecal specimens sampled from porcine G5P[7] K71 strain-inoculated calves were also examined by real-time RT-PCR. Note that viral RNA copy numbers in the feces from virus-inoculated calves were markedly lower than those from virus-inoculated piglets. (B and C) The sequential changes of viral RNA copy numbers in the feces, serum, MLN, liver, lung, and choroid plexus sampled from virus-inoculated piglets (B) or calves (C) were similar to each other. The geometric means of virus RNA copy numbers per mg of each sample are displayed.
| 과제명(ProjectTitle) : | - |
|---|---|
| 연구책임자(Manager) : | - |
| 과제기간(DetailSeriesProject) : | - |
| 총연구비 (DetailSeriesProject) : | - |
| 키워드(keyword) : | - |
| 과제수행기간(LeadAgency) : | - |
| 연구목표(Goal) : | - |
| 연구내용(Abstract) : | - |
| 기대효과(Effect) : | - |
Copyright KISTI. All Rights Reserved.

※ AI-Helper는 부적절한 답변을 할 수 있습니다.