최소 단어 이상 선택하여야 합니다.
최대 10 단어까지만 선택 가능합니다.
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
NTIS 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
DataON 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Edison 바로가기
다음과 같은 기능을 한번의 로그인으로 사용 할 수 있습니다.
Kafe 바로가기
| 주관연구기관 | 중앙대학교 Chung Ang University |
|---|---|
| 연구책임자 | 김홍진 |
| 참여연구자 | 이덕찬 , 강보규 |
| 보고서유형 | 최종보고서 |
| 발행국가 | 대한민국 |
| 언어 | 한국어 |
| 발행년월 | 2018-08 |
| 주관부처 | 해양수산부 Ministry of Oceans and Fisheries |
| 등록번호 | TRKO201900000120 |
| DB 구축일자 | 2019-06-01 |
| 키워드 | 노다바이러스.재조합 백신.바이러스 유사입자.경구용 백신.효모.Nodavirus.recombinant vaccine.virus-like particle.oral vaccine.yeast. |
본 연구에서는 바이러스 유사입자를 기반으로 한 재조합 노다바이러스 주사용 및 경구용 백신을 개발하였고 생산된 노다바이러스 백신 시제품을 양식 현장 어체에 적용하여 백신 효능을 입증하였음.
(출처 : 요약서 3p)
Purpose & Contents
Purpose
1. Development of recombinant nodavirus vaccine based on virus-like particle (VLP)
2. Production of trial product of recombinant nodavirus vaccine as an injectable vaccine, which is less expensive and highly efficient, and field trial in the fish cultivation
3.
Purpose & Contents
Purpose
1. Development of recombinant nodavirus vaccine based on virus-like particle (VLP)
2. Production of trial product of recombinant nodavirus vaccine as an injectable vaccine, which is less expensive and highly efficient, and field trial in the fish cultivation
3. Production of trial product of recombinant yeasts expressing nodavirus capsid protein as an oral vaccine, which is less expensive and highly efficient, and field trial of the trial product in the fish cultivation
Contents
1. Establishment of highly efficient production condition for recombinant nodavirus vaccine antigen and production condition for maximizing immunogenicity of the vaccine antigen.
2. Establishments of standard operating procedures for large-scale productions of recombinant nodavirus vaccines.
3. Verification of stability and safety of recombinant nodavirus vaccine.
4. Efficacy test against recombinant nodavirus vaccine (including field trial in fish cultivation).
Results
1. Development of cell lines for producing vaccine antigens (oral and injectable vaccine)
- Yeast cell lines for producing vaccine antigens were established.
- Expression stabilities of the antigens in the yeast cell lines when the passage number increased were confirmed.
2. Establishments of systems for large scale yeast fermentation (oral and injectable vaccines)
- Culture condition of yeast cells which is potentially usable for large scale fermentation was established.
- Condition for purifying the antigens for injectable vaccine was established, and the content was published as article and patent.
3. Establishments of fish systems to investigate the vaccine efficacy (oral and injectable vaccine).
- The vaccine efficacies were verified in convict grouper and olive flounder model.
- In the convict grouper model, the vaccine efficacies were verified as presences of serum antibodies against nodavirus capsid protein following vaccination, and survival rates post nodavirus challenge to the vaccinated fish were checked.
- In olive flounder model, the vaccine efficacies were verified as generations of the serum antibodies against nodavirus capsid protein.
4. Verifications of stabilities and safeties of injectable and oral vaccines
- The stabilities of vaccine antigens were verified in various conditions (the condition for preserving antigen was varied and the vaccine antigens were exposed to unfavorable conditions), and optimum conditions for preserving the antigen’s properties were established. Corresponding contents were published as article.
5. Establishments of standard operating procedures for producing injectable and oral vaccines
- The standard operating procedures for vaccine productions were established, and stabilities in antigen productions were confirmed from three production batches when it was conducted according to the operating procedures.
6. Suggestion of parameters for evaluating vaccination programs (injectable and oral vaccines)
- The guidelines for evaluating antibody generations following vaccination program were established.
7. Development of optimized formulation for oral vaccine
- Vaccine formulation to increase voluntary consumption of fish were established.
8. Productions of prototype vaccines (oral and injectable vaccines)
- The prototype vaccines were manufactured, and their abilities to elicit antibodies were evaluated.
9. Verifications of efficacies of injectable and oral vaccines (field trials to fish hatchery)
- The generations of antibodies and protective immunities against nodavirus following vaccinations were verified in convict grouper. Corresponding contents were published in article (SCI; top 10% of journal)
10. Studies for improving manufacturing processes which reduce production costs of injectable and oral vaccines were conducted.
- The cell culture condition for reducing use of galactose was selected. It was confirmed that the changed culture condition satisfies the yield of vaccine antigen we aimed.
11. Assessment of sensitivity of olive flounder to nodavirus infection
- The pathogenicity of nodavirus to olive flounder was investigated to assess the applicability of the olive flounder model as verifying the vaccine efficacy.
12. Investigation of immune response of vaccine following use of adjuvant
- The adjuvant was applied to reduce the amount of vaccine antigen, and the antibody responses as a function of the adjuvant were investigated.
Expected Contribution
1. Improvement of constitution in domestic fish cultivation area and enhancement of farm fish yield will be possible through providing nodavirus vaccine.
2. Aquisition of international competitiveness to export farm fish will be possible.
3. Aquisition of technical superiority to control viral disease of fish in international research field through development of recombinant virus vaccine for fish will be possible.
(출처 : SUMMARY 6p)
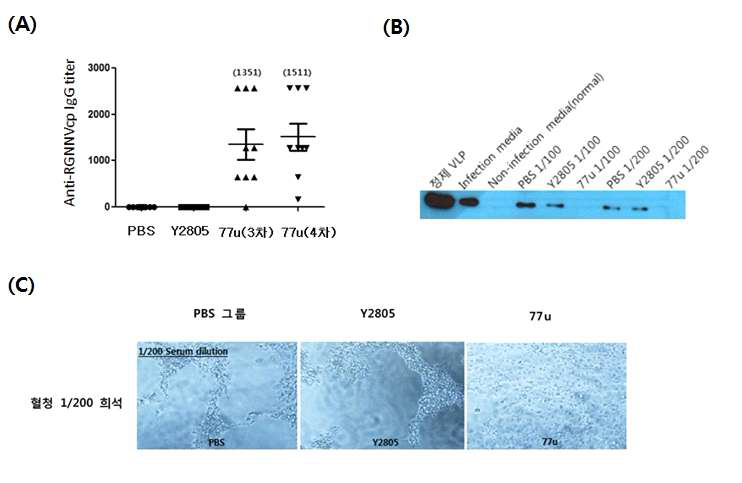 표
산업화 가능 균주(77u, GRAS 등급)의 효능 확인. A, 산업화 가능 균주의 항체 유도능 테스트. 77u를 3회, 4회 마우스에 경구 면역한 후 유도된 항체 역가를 ELISA를 통해 확인하였음. PBS는 생리식염수 투여군, Y2805는 77u의 parent 세포주를 의미함; B, 각 그룹의 마우스 혈청을 RGNNV와 mix한후 E-11 세포에 감염시킨 후 생성된 RGNNV의 캡시드 단백질 발현양을 Western blot으로 확인한 결과 77u로 면역한 마우스 혈청이 RGNNV의 증식을 효과적으로 차단함을 확인하였음. C, 각 그룹의 혈청을 RGNNV와 mix한 후 세포에 감염시키고 CPE를 관찰한 결과 77u로 면역한 마우스 혈청이 CPE형성을 효과적으로 차단함을 확인하였음
표
산업화 가능 균주(77u, GRAS 등급)의 효능 확인. A, 산업화 가능 균주의 항체 유도능 테스트. 77u를 3회, 4회 마우스에 경구 면역한 후 유도된 항체 역가를 ELISA를 통해 확인하였음. PBS는 생리식염수 투여군, Y2805는 77u의 parent 세포주를 의미함; B, 각 그룹의 마우스 혈청을 RGNNV와 mix한후 E-11 세포에 감염시킨 후 생성된 RGNNV의 캡시드 단백질 발현양을 Western blot으로 확인한 결과 77u로 면역한 마우스 혈청이 RGNNV의 증식을 효과적으로 차단함을 확인하였음. C, 각 그룹의 혈청을 RGNNV와 mix한 후 세포에 감염시키고 CPE를 관찰한 결과 77u로 면역한 마우스 혈청이 CPE형성을 효과적으로 차단함을 확인하였음
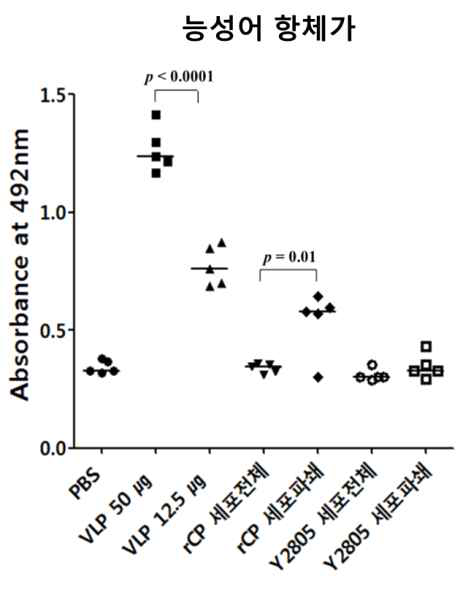 표
능성어 모델에서 백신 접종 후 RGNNV capsid에 대한 항체 수준 측정 결과. 능성어 모델에서 주사용 백신과 경구용 백신을 접종 한 그룹의 RGNNV capsid protein에 대한 혈중 항체를 ELISA를 통해 측정하였음. 주사용 백신 그룹을 VLP 50과 VLP 12.5 그룹으로 나누었고 각각 50 ㎍, 12.5 ㎍의 VLP를 1회 주사하였음. PBS를 주사한 그룹을 대조군으로 설정하였음. 경구용 백신의 경우 백신 효능 측정 그룹을 rCP 세포전체와 rCP 세포파쇄로 나누었고 각각 50 mg의 VLP를 세포전체(rCP 세포 전체)로 경구투여 하거나 세포파쇄 후 dialysis 진행 한 제형(rCP 세포 파쇄)을 경구투여 하였음. Y2805 그룹은 경구 백신의 대조군으로 설정하였고 Y2805 세포전체와 Y2805 세포파쇄 그룹으로 나누어 투여하였음
표
능성어 모델에서 백신 접종 후 RGNNV capsid에 대한 항체 수준 측정 결과. 능성어 모델에서 주사용 백신과 경구용 백신을 접종 한 그룹의 RGNNV capsid protein에 대한 혈중 항체를 ELISA를 통해 측정하였음. 주사용 백신 그룹을 VLP 50과 VLP 12.5 그룹으로 나누었고 각각 50 ㎍, 12.5 ㎍의 VLP를 1회 주사하였음. PBS를 주사한 그룹을 대조군으로 설정하였음. 경구용 백신의 경우 백신 효능 측정 그룹을 rCP 세포전체와 rCP 세포파쇄로 나누었고 각각 50 mg의 VLP를 세포전체(rCP 세포 전체)로 경구투여 하거나 세포파쇄 후 dialysis 진행 한 제형(rCP 세포 파쇄)을 경구투여 하였음. Y2805 그룹은 경구 백신의 대조군으로 설정하였고 Y2805 세포전체와 Y2805 세포파쇄 그룹으로 나누어 투여하였음
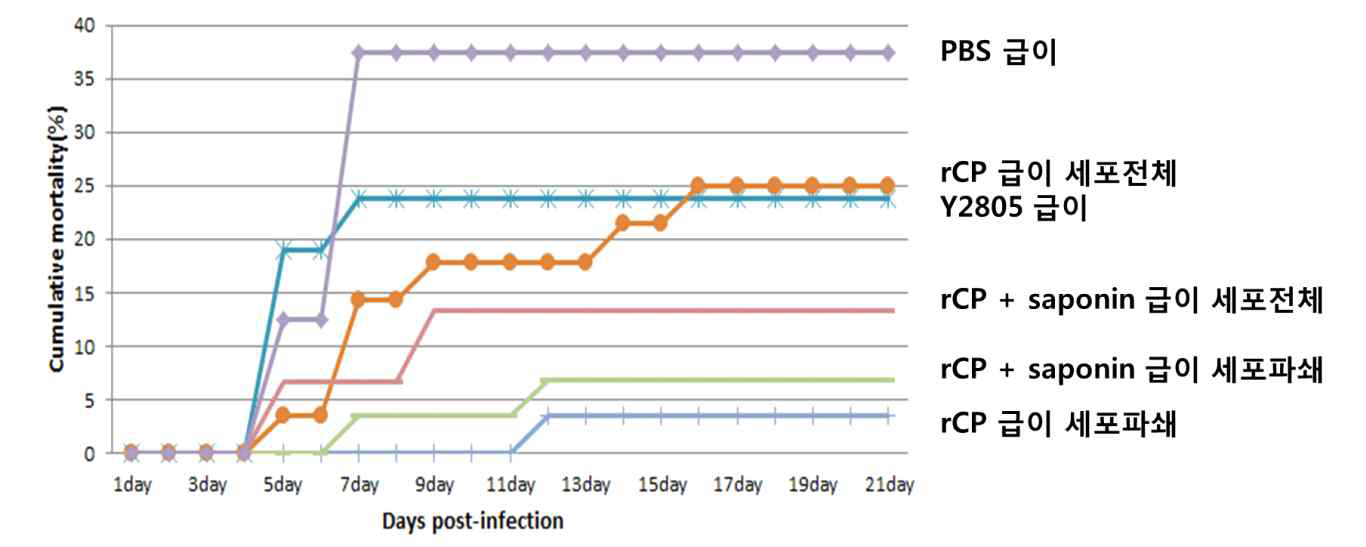 표
능성어 모델에서 경구용 백신 제조방법 (효모파쇄여부) 및 사포닌 첨가에 따른 RGNNV 인위감염 누적 폐사율. 경구용 백신 항원 rCP를 발현하는 효모의 파쇄 및 비파쇄 형태를 각각 동결건조하여 백신 실험에 사용하였음. 경구용 백신 실험에서 사포닌이 면역보조제로의 사용여부를 알아보기 위하여 동결건조된 rCP발현 효모와 함께 투여하였음. 능성어 (16.4cm±1.32cm,70.3±17.27gram)를 25도에서 1주일간 순치 후 경구용 백신을 일주일 간격으로 4회 투여하였으며 1차 백신 접종 후 41일째 능성어에서 혈청을 확보하였음. 1차 백신 접종 후 47일 째에 RGNNV를 감염시켰고 바이러스 감염에 따른 능성어의 누적 폐사율을 관찰하였음
표
능성어 모델에서 경구용 백신 제조방법 (효모파쇄여부) 및 사포닌 첨가에 따른 RGNNV 인위감염 누적 폐사율. 경구용 백신 항원 rCP를 발현하는 효모의 파쇄 및 비파쇄 형태를 각각 동결건조하여 백신 실험에 사용하였음. 경구용 백신 실험에서 사포닌이 면역보조제로의 사용여부를 알아보기 위하여 동결건조된 rCP발현 효모와 함께 투여하였음. 능성어 (16.4cm±1.32cm,70.3±17.27gram)를 25도에서 1주일간 순치 후 경구용 백신을 일주일 간격으로 4회 투여하였으며 1차 백신 접종 후 41일째 능성어에서 혈청을 확보하였음. 1차 백신 접종 후 47일 째에 RGNNV를 감염시켰고 바이러스 감염에 따른 능성어의 누적 폐사율을 관찰하였음
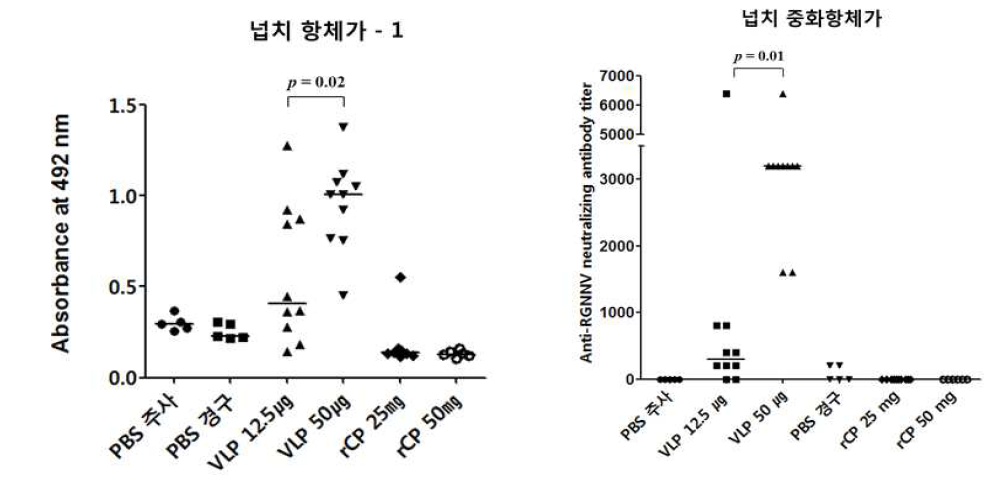 표
넙치 모델에서 주사용 백신과 경구용 백신의 접종에 따른 RGNNV capsid protein에 대한 혈중 항체 역가 및 RGNNV에 대한 혈중 중화항체가 측정 결과. RGNNV capsid에 대한 혈중 항체가는 ELISA를 통해 측정되었고 RGNNV에 대한 혈중 중화항체가는 E-11 세포기반 방법을 통해 CPE 형성 유무를 통해 확인하였음. 실험그룹은 주사용 백신그룹과 경구용 백신그룹으로 나누었고 대조군으로 각각 PBS 주사 그룹과 PBS 경구 그룹으로 설정하였음. 주사용 백신의 접종량은 dose당 12.5 ㎍과 50 ㎍ 두 종류로 나누었고 경구용 백신의 접종량은 25 mg과 50 mg으로 나누었음
표
넙치 모델에서 주사용 백신과 경구용 백신의 접종에 따른 RGNNV capsid protein에 대한 혈중 항체 역가 및 RGNNV에 대한 혈중 중화항체가 측정 결과. RGNNV capsid에 대한 혈중 항체가는 ELISA를 통해 측정되었고 RGNNV에 대한 혈중 중화항체가는 E-11 세포기반 방법을 통해 CPE 형성 유무를 통해 확인하였음. 실험그룹은 주사용 백신그룹과 경구용 백신그룹으로 나누었고 대조군으로 각각 PBS 주사 그룹과 PBS 경구 그룹으로 설정하였음. 주사용 백신의 접종량은 dose당 12.5 ㎍과 50 ㎍ 두 종류로 나누었고 경구용 백신의 접종량은 25 mg과 50 mg으로 나누었음
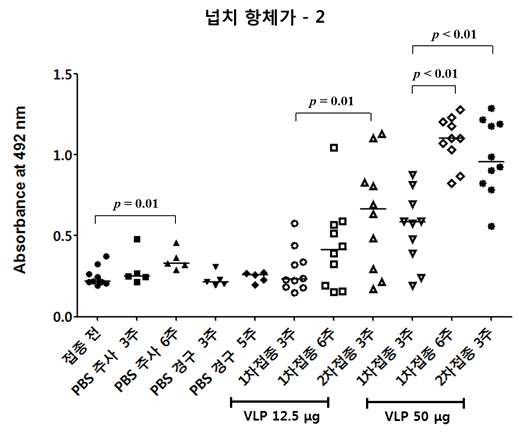 표
넙치 모델에서 주사용 백신 접종 후 항체가 측정 결과. 본 결과는 접종 횟수 및 접종 후 혈액 채취 시간에 따른 각 그룹의 RGNNV capsid protein에 대한 혈중 항체가 측정 결과를 보여줌. RGNNV capsid에 대한 혈중 항체가는 ELISA를 통해 측정되었음. 주사용 백신의 접종량은 12.5 ㎍과 50 ㎍ 두 종류로 나누었고 백신 접종 후 혈액 채취 시간을 각각 1차 접종 후 3주, 1차 접종 후 6주, 2차 접종 후 3주로 나누었음. 대조군으로 접종 전, PBS 주사 후 3주, PBS 주사 후 6주, PBS 경구 투여 후 3주, PBS 경구 투여 후 5주 그룹으로 설정하였음
표
넙치 모델에서 주사용 백신 접종 후 항체가 측정 결과. 본 결과는 접종 횟수 및 접종 후 혈액 채취 시간에 따른 각 그룹의 RGNNV capsid protein에 대한 혈중 항체가 측정 결과를 보여줌. RGNNV capsid에 대한 혈중 항체가는 ELISA를 통해 측정되었음. 주사용 백신의 접종량은 12.5 ㎍과 50 ㎍ 두 종류로 나누었고 백신 접종 후 혈액 채취 시간을 각각 1차 접종 후 3주, 1차 접종 후 6주, 2차 접종 후 3주로 나누었음. 대조군으로 접종 전, PBS 주사 후 3주, PBS 주사 후 6주, PBS 경구 투여 후 3주, PBS 경구 투여 후 5주 그룹으로 설정하였음
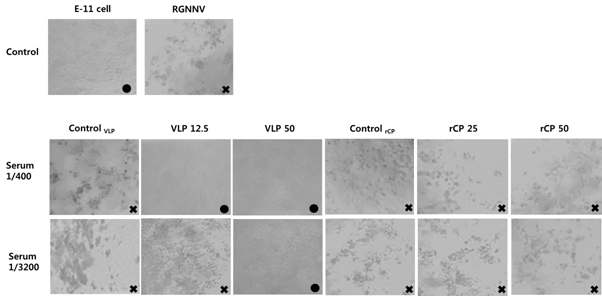 표
넙치 모델에서 주사용, 경구용 백신 투여에 따른 중화활성 유도 확인 결과. 넙치에 주사용, 경구용 백신을 표 9 및 표 10과 같이 투여한 후 주사용 백신 2차 접종 3주후, 경구용 백신 4차 투여 3주후에 혈청을 취하고, 혈청 내 RGNNV 중화활성을 E-11 세포에 대한 감염시험을 통해 확인하였음. 각 그룹의 혈액은 각각 1/400과 1/3200로 희석하였음. Control 결과에서 ‘E-11 cell’과 ‘RGNNV’는 RGNNV를 감염시키지 않는 정상적인 세포와 RGNNV를 감염시킨 세포 상태를 각각 보여줌. Control VLP는 PBS를 주사로 투여한 그룹의 혈청 적용 결과를 보여줌. VLP 12.5와 VLP 50은 각각 RGNNV 주사용 백신 12.5 μg 투여그룹과 50 μg 투여그룹의 혈청 적용 결과를 보여줌. Control rCP는 경구백신에 대한 대조군으로 PBS를 경구로 투여한 그룹의 혈청 적용 결과를 보여줌. rCP 25와 rCP 50은 dose당 25 mg과 50 mg을 각각 4회씩 경구로 투여한 그룹의 혈청 적용 결과를 보여줌
표
넙치 모델에서 주사용, 경구용 백신 투여에 따른 중화활성 유도 확인 결과. 넙치에 주사용, 경구용 백신을 표 9 및 표 10과 같이 투여한 후 주사용 백신 2차 접종 3주후, 경구용 백신 4차 투여 3주후에 혈청을 취하고, 혈청 내 RGNNV 중화활성을 E-11 세포에 대한 감염시험을 통해 확인하였음. 각 그룹의 혈액은 각각 1/400과 1/3200로 희석하였음. Control 결과에서 ‘E-11 cell’과 ‘RGNNV’는 RGNNV를 감염시키지 않는 정상적인 세포와 RGNNV를 감염시킨 세포 상태를 각각 보여줌. Control VLP는 PBS를 주사로 투여한 그룹의 혈청 적용 결과를 보여줌. VLP 12.5와 VLP 50은 각각 RGNNV 주사용 백신 12.5 μg 투여그룹과 50 μg 투여그룹의 혈청 적용 결과를 보여줌. Control rCP는 경구백신에 대한 대조군으로 PBS를 경구로 투여한 그룹의 혈청 적용 결과를 보여줌. rCP 25와 rCP 50은 dose당 25 mg과 50 mg을 각각 4회씩 경구로 투여한 그룹의 혈청 적용 결과를 보여줌
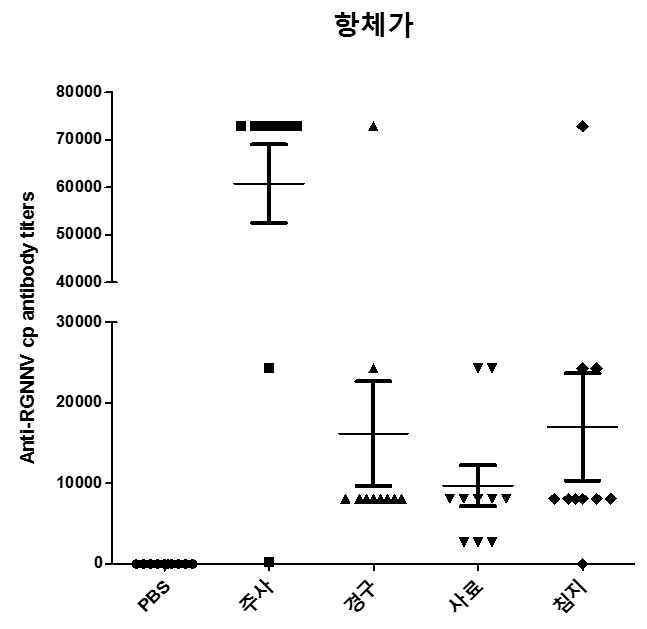 표
노다바이러스 주사용 백신 및 경구용 백신의 투여에 따른 능성어 혈중 항 -RGNNV capsid protein 항체 수준 측정결과. 수집한 혈액을 생리식염수에 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 skim milk로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체 -RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. control, n=10. 주사, n=10. 경구, n=10. 사료, n=10. 침지, n=10
표
노다바이러스 주사용 백신 및 경구용 백신의 투여에 따른 능성어 혈중 항 -RGNNV capsid protein 항체 수준 측정결과. 수집한 혈액을 생리식염수에 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 skim milk로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체 -RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. control, n=10. 주사, n=10. 경구, n=10. 사료, n=10. 침지, n=10
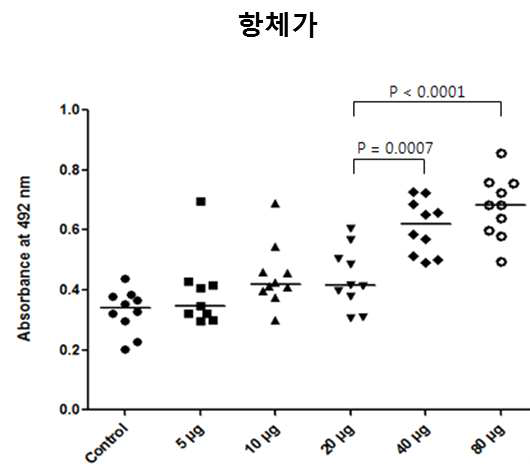 표
노다바이러스 주사용 백신의 접종에 따른 능성어 혈 중 항-RGNNV capsid protein 항체 수준을 ELISA기법으로 측정한 결과. 수집한 혈액을 생리식염수에 1/400 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 BSA로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체 -RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 492 nm에서 OPD 발색 시약의 발색정도로 나타내었음. control, n=10. 5 μg, n=10. 10 μg, n=10. 20 μg, n=10. 40 μg, n=10. 80 μg, n=10
표
노다바이러스 주사용 백신의 접종에 따른 능성어 혈 중 항-RGNNV capsid protein 항체 수준을 ELISA기법으로 측정한 결과. 수집한 혈액을 생리식염수에 1/400 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 BSA로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체 -RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 492 nm에서 OPD 발색 시약의 발색정도로 나타내었음. control, n=10. 5 μg, n=10. 10 μg, n=10. 20 μg, n=10. 40 μg, n=10. 80 μg, n=10
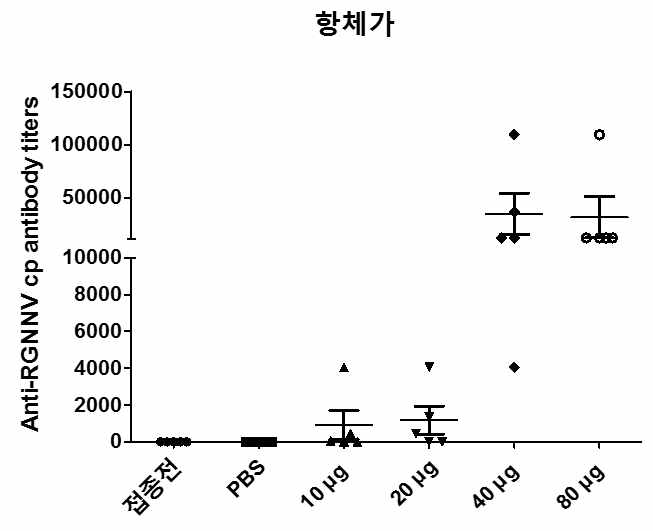 표
RGNNV 주사용 백신의 접종량에 따른 넙치 혈 중 항-RGNNV capsid protein 항체 수준을 ELISA 기법으로 측정한 결과. 수집한 혈액을 생리식염수에 three-fold로 연속 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 skim milk로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well 로 분주함으로서 어체 혈중 항체-RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. 접종 전 n=5. PBS, n=5. 10 μg, n=5. 20 μg, n=5. 40 μg, n=5. 80 μg, n=5
표
RGNNV 주사용 백신의 접종량에 따른 넙치 혈 중 항-RGNNV capsid protein 항체 수준을 ELISA 기법으로 측정한 결과. 수집한 혈액을 생리식염수에 three-fold로 연속 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 skim milk로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well 로 분주함으로서 어체 혈중 항체-RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. 접종 전 n=5. PBS, n=5. 10 μg, n=5. 20 μg, n=5. 40 μg, n=5. 80 μg, n=5
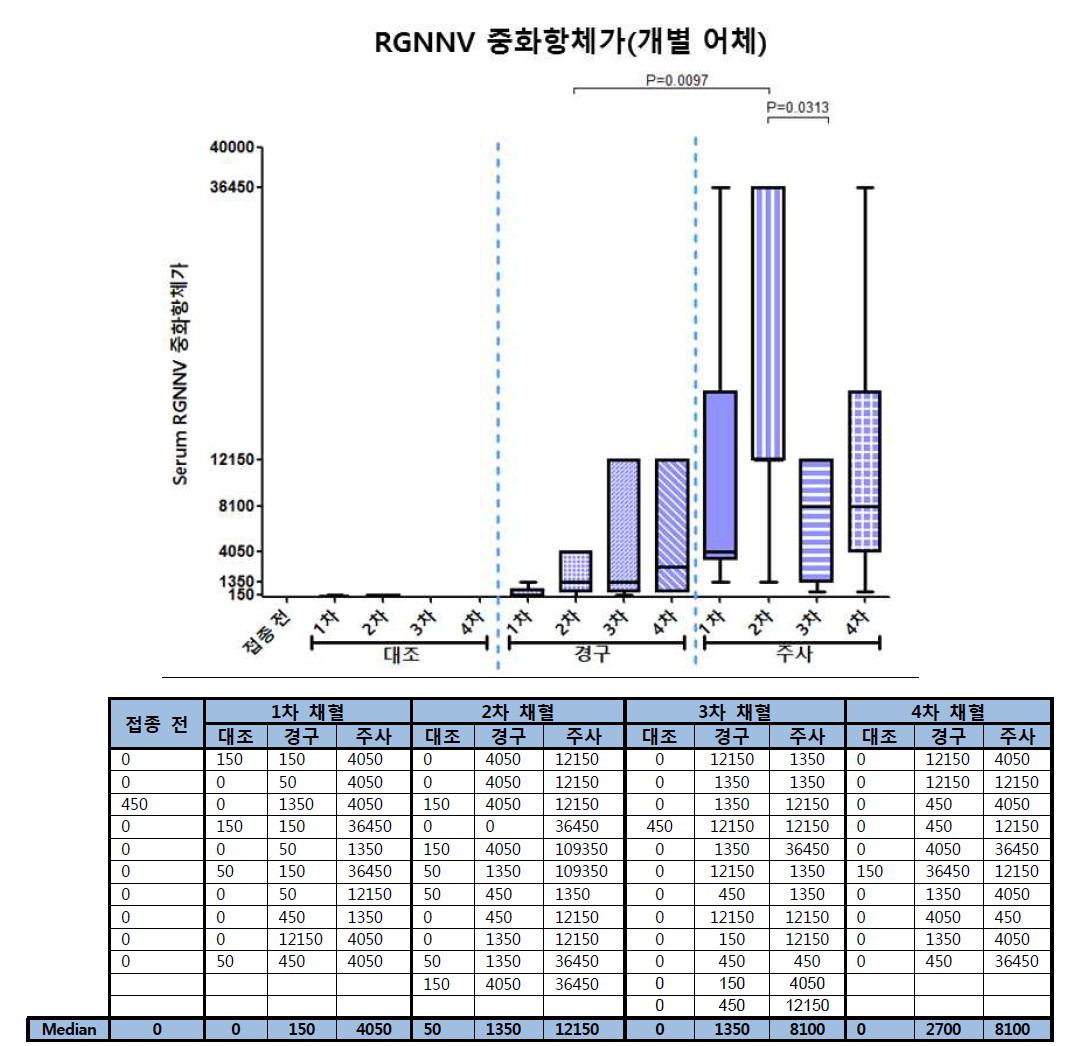 표
노다바이러스 주사 백신 및 경구 백신의 투여에 따른 능성어 혈 중 항-노다바이러스 중화항체 역가 측정결과. 그룹당 개별 어체 혈액을 1/50부터 1/109,350까지 3배 희석배율로 희석하여 RGNNV 바이러스와 혼합한 다음 E-11세포에 분주하였음. 세포병변이 관찰되지 않는 혈액의 최고 희석비율을 serum 중화항체 역가로 정하였음. 결과에서 그룹 당 25%-75% 값의 범위를 박스로 나타내었고 median값을 선으로 나타내었음. 접종전, n=10. 1차채혈(대조, 경구, 주사), n=10. 2차채혈(대조, 경구, 주사), n=11. 3차채혈(대조, 경구, 주사), n=12. 4차채혈(대조, 경구, 주사), n=10
표
노다바이러스 주사 백신 및 경구 백신의 투여에 따른 능성어 혈 중 항-노다바이러스 중화항체 역가 측정결과. 그룹당 개별 어체 혈액을 1/50부터 1/109,350까지 3배 희석배율로 희석하여 RGNNV 바이러스와 혼합한 다음 E-11세포에 분주하였음. 세포병변이 관찰되지 않는 혈액의 최고 희석비율을 serum 중화항체 역가로 정하였음. 결과에서 그룹 당 25%-75% 값의 범위를 박스로 나타내었고 median값을 선으로 나타내었음. 접종전, n=10. 1차채혈(대조, 경구, 주사), n=10. 2차채혈(대조, 경구, 주사), n=11. 3차채혈(대조, 경구, 주사), n=12. 4차채혈(대조, 경구, 주사), n=10
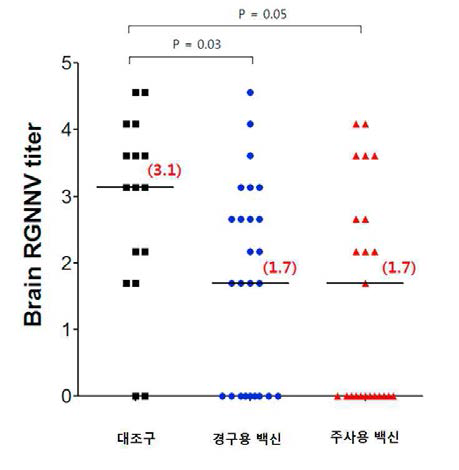 표
RGNNV 공격시험에서 경구백신 및 주사백신의 투여에 따른 능성어 뇌 조직의 RGNNV titer 측정 결과. 능성어 마리 당 1X106 TCID50 RGNNV를 challenge하였고 virus challenge 후 2주 뒤에 뇌 조직을 회수하였음. 회수한 뇌 조직을 각각 1/200부터 1/25,600 까지 2배 배율로 희석하여 E-11세포에 분주하였음. 세포병변이 관찰되는 뇌 조직 mixture의 최고 희석 비율을 brain RGNNV titer로 정하였음. 결과는 각 그룹의 brand RGNNV titer를 log10 스케일로 설정하여 median 값으로 나타내었음. 대조, n=16. 경구, n=25. 주사, n=25
표
RGNNV 공격시험에서 경구백신 및 주사백신의 투여에 따른 능성어 뇌 조직의 RGNNV titer 측정 결과. 능성어 마리 당 1X106 TCID50 RGNNV를 challenge하였고 virus challenge 후 2주 뒤에 뇌 조직을 회수하였음. 회수한 뇌 조직을 각각 1/200부터 1/25,600 까지 2배 배율로 희석하여 E-11세포에 분주하였음. 세포병변이 관찰되는 뇌 조직 mixture의 최고 희석 비율을 brain RGNNV titer로 정하였음. 결과는 각 그룹의 brand RGNNV titer를 log10 스케일로 설정하여 median 값으로 나타내었음. 대조, n=16. 경구, n=25. 주사, n=25
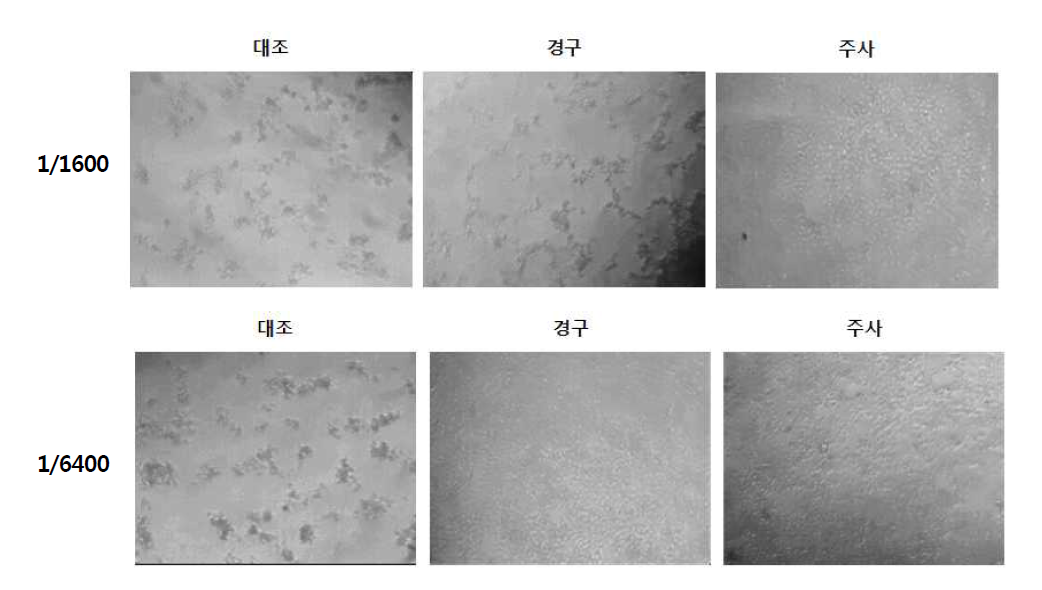 표
능성어 뇌 조직내 RGNNV titer 측정 시 E-11세포에 대한 세포병변을 현미경으로 관찰한 결과. 능성어 마리 당 1X106 TCID50 RGNNV를 challenge하였고 virus challenge 후 2주 뒤에 뇌 조직을 회수하였음. 각 그룹당 뇌 조직을 혼합하여 mixture를 만들고 mixture를 각각 1/200부터 1/25,600 까지 2배 배율로 희석하여 E-11세포에 분주하여 세포병변을 관찰하였음. 1/1,600과 1/6,400 비율에서 관찰한 현미경 결과를 대표결과로 나타내었음. 대조, n=16. 경구, n=25. 주사, n=25
표
능성어 뇌 조직내 RGNNV titer 측정 시 E-11세포에 대한 세포병변을 현미경으로 관찰한 결과. 능성어 마리 당 1X106 TCID50 RGNNV를 challenge하였고 virus challenge 후 2주 뒤에 뇌 조직을 회수하였음. 각 그룹당 뇌 조직을 혼합하여 mixture를 만들고 mixture를 각각 1/200부터 1/25,600 까지 2배 배율로 희석하여 E-11세포에 분주하여 세포병변을 관찰하였음. 1/1,600과 1/6,400 비율에서 관찰한 현미경 결과를 대표결과로 나타내었음. 대조, n=16. 경구, n=25. 주사, n=25
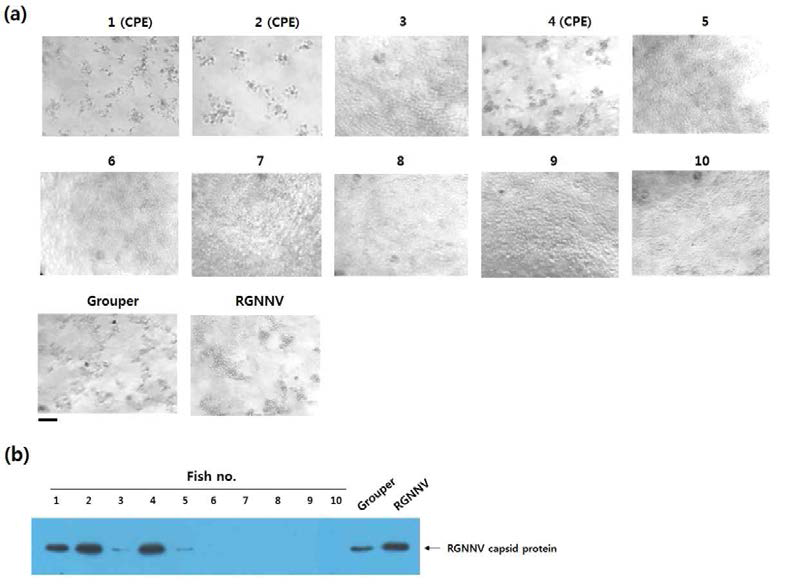 표
시중 유통 넙치 brain의 RGNNV 활성도 조사 결과. 넙치 brain extract를 E-11 세포에 적용한 후 8일간 배양하고 CPE 형성 유무를 현미경 관찰을 통해 확인하였음(A). 각 culture의 세포 및 배양액 내 RGNNV capsid protein의 유무를 Western blot으로 확인하였음(B). CPE가 관찰된 배양 well은 ‘CPE’로 표기함. Groper는 106 TCID50으로 RGNNV를 능성어에 challenge한 후 brain extract를 적용한 well을 의미함. RGNNV는 E-11에 200 TCID50의 RGNNV를 직접 적용한 well을 의미함(positive control). Bar,50μm
표
시중 유통 넙치 brain의 RGNNV 활성도 조사 결과. 넙치 brain extract를 E-11 세포에 적용한 후 8일간 배양하고 CPE 형성 유무를 현미경 관찰을 통해 확인하였음(A). 각 culture의 세포 및 배양액 내 RGNNV capsid protein의 유무를 Western blot으로 확인하였음(B). CPE가 관찰된 배양 well은 ‘CPE’로 표기함. Groper는 106 TCID50으로 RGNNV를 능성어에 challenge한 후 brain extract를 적용한 well을 의미함. RGNNV는 E-11에 200 TCID50의 RGNNV를 직접 적용한 well을 의미함(positive control). Bar,50μm
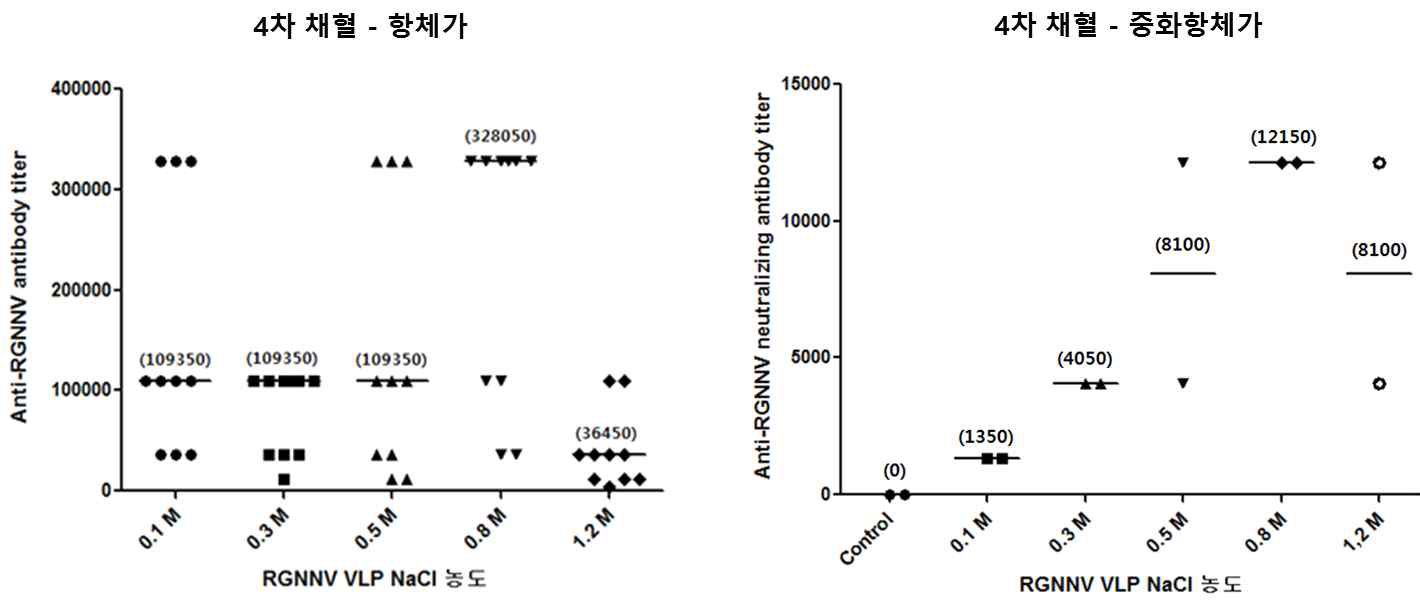 표
정제된 RGNNV VLP의 준비 용액에 따른 마우스 혈 중 항체 역가 및 중화항체 역가 측정결과. 정제된 RGNNV VLP를 각각 0.1, 0.3, 0.5, 0.8, 1.2 M NaCl을 포함한 TBST에서 투석하여 준비함. 조건별로 준비한 RGNNV VLP 항원을 Balb/c 마우스에 100 ng/dose, 2주 간격으로 총 4회 피하주사 진행하고 마지막 면역으로부터 10일 뒤 혈액을 수집하였음. 면역을 위해 별도의 면역보조제는 사용하지 않았음. 생리식염수를 주사한 마우스를 대조군으로 설정하였음. 마우스 혈 중 항-RGNNV capsid protein 항체 역가(왼쪽)는 ELISA기법으로 측정하였고 혈 중 RGNNV 중화항체 역가(오른쪽)는 E-11세포를 기반 중화항체 역가 측정법으로 측정하였음. 0.1 M NaCl, n=10. 0.3 M NaCl, n=10. 0.5 M NaCl, n=10. 0.8 M NaCl, n=10. 1.0 M NaCl, n=10
표
정제된 RGNNV VLP의 준비 용액에 따른 마우스 혈 중 항체 역가 및 중화항체 역가 측정결과. 정제된 RGNNV VLP를 각각 0.1, 0.3, 0.5, 0.8, 1.2 M NaCl을 포함한 TBST에서 투석하여 준비함. 조건별로 준비한 RGNNV VLP 항원을 Balb/c 마우스에 100 ng/dose, 2주 간격으로 총 4회 피하주사 진행하고 마지막 면역으로부터 10일 뒤 혈액을 수집하였음. 면역을 위해 별도의 면역보조제는 사용하지 않았음. 생리식염수를 주사한 마우스를 대조군으로 설정하였음. 마우스 혈 중 항-RGNNV capsid protein 항체 역가(왼쪽)는 ELISA기법으로 측정하였고 혈 중 RGNNV 중화항체 역가(오른쪽)는 E-11세포를 기반 중화항체 역가 측정법으로 측정하였음. 0.1 M NaCl, n=10. 0.3 M NaCl, n=10. 0.5 M NaCl, n=10. 0.8 M NaCl, n=10. 1.0 M NaCl, n=10
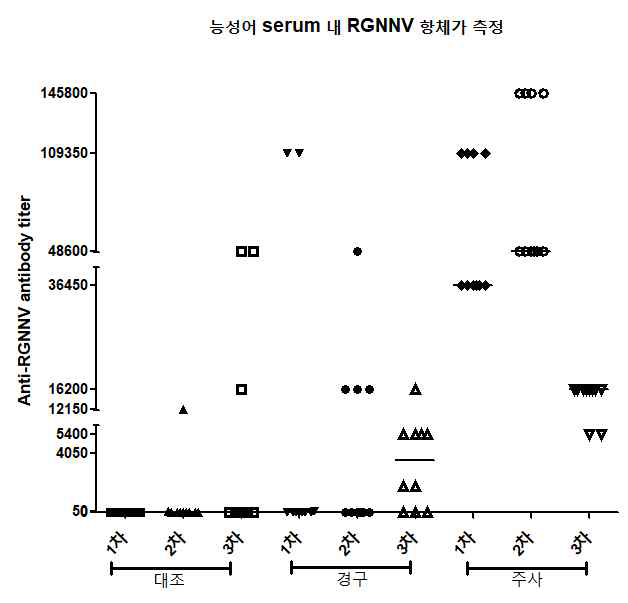 표
경구용 및 주사용 백신 접종 능성어의 1차, 2차, 3차 채혈 혈액의 항체 역가 측정 결과. 수집한 혈액을 각각 생리식염수에 희석하여 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 BSA로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체-RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. 대조, n=10. 경구, n=10. 주사, n=10
표
경구용 및 주사용 백신 접종 능성어의 1차, 2차, 3차 채혈 혈액의 항체 역가 측정 결과. 수집한 혈액을 각각 생리식염수에 희석하여 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 BSA로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체-RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. 대조, n=10. 경구, n=10. 주사, n=10
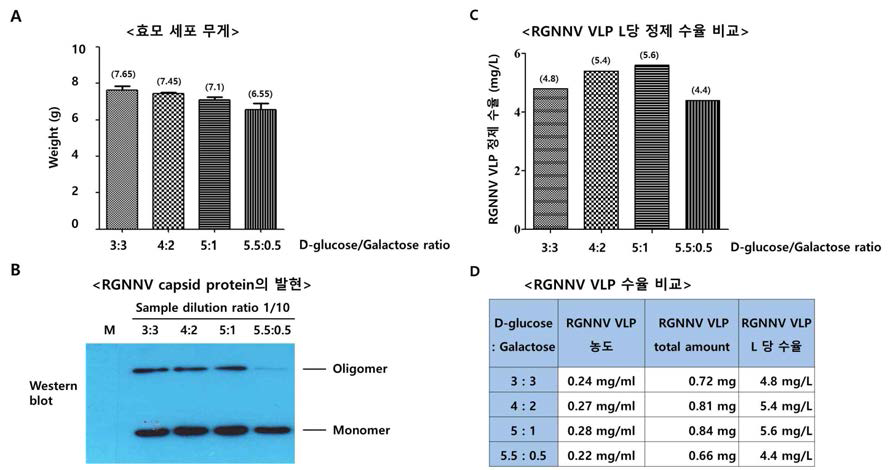 표
탄소원 배양 조건에 따른 주사용 백신 항원 발현 세포의 성장, 백신 항원 발현 및 항원 수율을 비교한 결과. 탄소원 6% 기준으로 D-glucose와 galactose를 각각 3:3, 4:, 5:1, 5.5:0.5 첨가하여 주사용 백신 항원 발현 효모 세포를 72 시간 동안 배양하고 A. 세포 무게를 측정함. B. 주사용 백신 항원의 발현을 Western blot으로 확인함. C. 배양한 효모 세포를 정제하여 최종 RGNNV VLP 주사용 백신 L당 수율을 확인함. D. 배양한 효모 세포를 정제하여 RGNNV VLP 주사용 백신 수율을 확인함
표
탄소원 배양 조건에 따른 주사용 백신 항원 발현 세포의 성장, 백신 항원 발현 및 항원 수율을 비교한 결과. 탄소원 6% 기준으로 D-glucose와 galactose를 각각 3:3, 4:, 5:1, 5.5:0.5 첨가하여 주사용 백신 항원 발현 효모 세포를 72 시간 동안 배양하고 A. 세포 무게를 측정함. B. 주사용 백신 항원의 발현을 Western blot으로 확인함. C. 배양한 효모 세포를 정제하여 최종 RGNNV VLP 주사용 백신 L당 수율을 확인함. D. 배양한 효모 세포를 정제하여 RGNNV VLP 주사용 백신 수율을 확인함
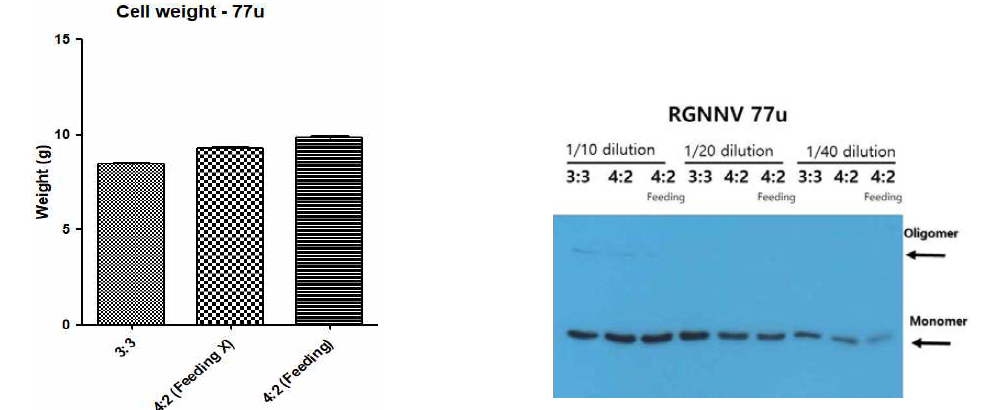 표
탄소원 배양 조건 및 feeding 조건에 따른 경구용 백신 항원 발현 세포의 성장률 및 백신 항원 발현양 확인한 결과. 탄소원 6% 기준으로 D-glucose와 galactose를 각각 3:3, 4:2로 조성하여 경구용 백신 항원 발현 효모 세포를 72 - 120시간 동안 배양함. 4:2(feeding) 조건의 경우 72 시간에 galactose를 1.5% 되게 첨가하고 120시간까지 배양하였음. 4:2 조건의 경우 feeding 없이 120시간 까지 배양하였고 3:3 조건의 경우 feeding 없이 72시간 까지 배양하였음. A. 최종 배양 후 세포 무게. B. 경구용 백신 항원의 발현을 Western blot으로 확인한 결과
표
탄소원 배양 조건 및 feeding 조건에 따른 경구용 백신 항원 발현 세포의 성장률 및 백신 항원 발현양 확인한 결과. 탄소원 6% 기준으로 D-glucose와 galactose를 각각 3:3, 4:2로 조성하여 경구용 백신 항원 발현 효모 세포를 72 - 120시간 동안 배양함. 4:2(feeding) 조건의 경우 72 시간에 galactose를 1.5% 되게 첨가하고 120시간까지 배양하였음. 4:2 조건의 경우 feeding 없이 120시간 까지 배양하였고 3:3 조건의 경우 feeding 없이 72시간 까지 배양하였음. A. 최종 배양 후 세포 무게. B. 경구용 백신 항원의 발현을 Western blot으로 확인한 결과
 표
산업화 가능 균주(77u, GRAS 등급)의 효능 확인. A, 산업화 가능 균주의 항체 유도능 테스트. 77u를 3회, 4회 마우스에 경구 면역한 후 유도된 항체 역가를 ELISA를 통해 확인하였음. PBS는 생리식염수 투여군, Y2805는 77u의 parent 세포주를 의미함; B, 각 그룹의 마우스 혈청을 RGNNV와 mix한후 E-11 세포에 감염시킨 후 생성된 RGNNV의 캡시드 단백질 발현양을 Western blot으로 확인한 결과 77u로 면역한 마우스 혈청이 RGNNV의 증식을 효과적으로 차단함을 확인하였음. C, 각 그룹의 혈청을 RGNNV와 mix한 후 세포에 감염시키고 CPE를 관찰한 결과 77u로 면역한 마우스 혈청이 CPE형성을 효과적으로 차단함을 확인하였음
표
산업화 가능 균주(77u, GRAS 등급)의 효능 확인. A, 산업화 가능 균주의 항체 유도능 테스트. 77u를 3회, 4회 마우스에 경구 면역한 후 유도된 항체 역가를 ELISA를 통해 확인하였음. PBS는 생리식염수 투여군, Y2805는 77u의 parent 세포주를 의미함; B, 각 그룹의 마우스 혈청을 RGNNV와 mix한후 E-11 세포에 감염시킨 후 생성된 RGNNV의 캡시드 단백질 발현양을 Western blot으로 확인한 결과 77u로 면역한 마우스 혈청이 RGNNV의 증식을 효과적으로 차단함을 확인하였음. C, 각 그룹의 혈청을 RGNNV와 mix한 후 세포에 감염시키고 CPE를 관찰한 결과 77u로 면역한 마우스 혈청이 CPE형성을 효과적으로 차단함을 확인하였음
 표
능성어 모델에서 백신 접종 후 RGNNV capsid에 대한 항체 수준 측정 결과. 능성어 모델에서 주사용 백신과 경구용 백신을 접종 한 그룹의 RGNNV capsid protein에 대한 혈중 항체를 ELISA를 통해 측정하였음. 주사용 백신 그룹을 VLP 50과 VLP 12.5 그룹으로 나누었고 각각 50 ㎍, 12.5 ㎍의 VLP를 1회 주사하였음. PBS를 주사한 그룹을 대조군으로 설정하였음. 경구용 백신의 경우 백신 효능 측정 그룹을 rCP 세포전체와 rCP 세포파쇄로 나누었고 각각 50 mg의 VLP를 세포전체(rCP 세포 전체)로 경구투여 하거나 세포파쇄 후 dialysis 진행 한 제형(rCP 세포 파쇄)을 경구투여 하였음. Y2805 그룹은 경구 백신의 대조군으로 설정하였고 Y2805 세포전체와 Y2805 세포파쇄 그룹으로 나누어 투여하였음
표
능성어 모델에서 백신 접종 후 RGNNV capsid에 대한 항체 수준 측정 결과. 능성어 모델에서 주사용 백신과 경구용 백신을 접종 한 그룹의 RGNNV capsid protein에 대한 혈중 항체를 ELISA를 통해 측정하였음. 주사용 백신 그룹을 VLP 50과 VLP 12.5 그룹으로 나누었고 각각 50 ㎍, 12.5 ㎍의 VLP를 1회 주사하였음. PBS를 주사한 그룹을 대조군으로 설정하였음. 경구용 백신의 경우 백신 효능 측정 그룹을 rCP 세포전체와 rCP 세포파쇄로 나누었고 각각 50 mg의 VLP를 세포전체(rCP 세포 전체)로 경구투여 하거나 세포파쇄 후 dialysis 진행 한 제형(rCP 세포 파쇄)을 경구투여 하였음. Y2805 그룹은 경구 백신의 대조군으로 설정하였고 Y2805 세포전체와 Y2805 세포파쇄 그룹으로 나누어 투여하였음
 표
능성어 모델에서 경구용 백신 제조방법 (효모파쇄여부) 및 사포닌 첨가에 따른 RGNNV 인위감염 누적 폐사율. 경구용 백신 항원 rCP를 발현하는 효모의 파쇄 및 비파쇄 형태를 각각 동결건조하여 백신 실험에 사용하였음. 경구용 백신 실험에서 사포닌이 면역보조제로의 사용여부를 알아보기 위하여 동결건조된 rCP발현 효모와 함께 투여하였음. 능성어 (16.4cm±1.32cm,70.3±17.27gram)를 25도에서 1주일간 순치 후 경구용 백신을 일주일 간격으로 4회 투여하였으며 1차 백신 접종 후 41일째 능성어에서 혈청을 확보하였음. 1차 백신 접종 후 47일 째에 RGNNV를 감염시켰고 바이러스 감염에 따른 능성어의 누적 폐사율을 관찰하였음
표
능성어 모델에서 경구용 백신 제조방법 (효모파쇄여부) 및 사포닌 첨가에 따른 RGNNV 인위감염 누적 폐사율. 경구용 백신 항원 rCP를 발현하는 효모의 파쇄 및 비파쇄 형태를 각각 동결건조하여 백신 실험에 사용하였음. 경구용 백신 실험에서 사포닌이 면역보조제로의 사용여부를 알아보기 위하여 동결건조된 rCP발현 효모와 함께 투여하였음. 능성어 (16.4cm±1.32cm,70.3±17.27gram)를 25도에서 1주일간 순치 후 경구용 백신을 일주일 간격으로 4회 투여하였으며 1차 백신 접종 후 41일째 능성어에서 혈청을 확보하였음. 1차 백신 접종 후 47일 째에 RGNNV를 감염시켰고 바이러스 감염에 따른 능성어의 누적 폐사율을 관찰하였음
 표
넙치 모델에서 주사용 백신과 경구용 백신의 접종에 따른 RGNNV capsid protein에 대한 혈중 항체 역가 및 RGNNV에 대한 혈중 중화항체가 측정 결과. RGNNV capsid에 대한 혈중 항체가는 ELISA를 통해 측정되었고 RGNNV에 대한 혈중 중화항체가는 E-11 세포기반 방법을 통해 CPE 형성 유무를 통해 확인하였음. 실험그룹은 주사용 백신그룹과 경구용 백신그룹으로 나누었고 대조군으로 각각 PBS 주사 그룹과 PBS 경구 그룹으로 설정하였음. 주사용 백신의 접종량은 dose당 12.5 ㎍과 50 ㎍ 두 종류로 나누었고 경구용 백신의 접종량은 25 mg과 50 mg으로 나누었음
표
넙치 모델에서 주사용 백신과 경구용 백신의 접종에 따른 RGNNV capsid protein에 대한 혈중 항체 역가 및 RGNNV에 대한 혈중 중화항체가 측정 결과. RGNNV capsid에 대한 혈중 항체가는 ELISA를 통해 측정되었고 RGNNV에 대한 혈중 중화항체가는 E-11 세포기반 방법을 통해 CPE 형성 유무를 통해 확인하였음. 실험그룹은 주사용 백신그룹과 경구용 백신그룹으로 나누었고 대조군으로 각각 PBS 주사 그룹과 PBS 경구 그룹으로 설정하였음. 주사용 백신의 접종량은 dose당 12.5 ㎍과 50 ㎍ 두 종류로 나누었고 경구용 백신의 접종량은 25 mg과 50 mg으로 나누었음
 표
넙치 모델에서 주사용 백신 접종 후 항체가 측정 결과. 본 결과는 접종 횟수 및 접종 후 혈액 채취 시간에 따른 각 그룹의 RGNNV capsid protein에 대한 혈중 항체가 측정 결과를 보여줌. RGNNV capsid에 대한 혈중 항체가는 ELISA를 통해 측정되었음. 주사용 백신의 접종량은 12.5 ㎍과 50 ㎍ 두 종류로 나누었고 백신 접종 후 혈액 채취 시간을 각각 1차 접종 후 3주, 1차 접종 후 6주, 2차 접종 후 3주로 나누었음. 대조군으로 접종 전, PBS 주사 후 3주, PBS 주사 후 6주, PBS 경구 투여 후 3주, PBS 경구 투여 후 5주 그룹으로 설정하였음
표
넙치 모델에서 주사용 백신 접종 후 항체가 측정 결과. 본 결과는 접종 횟수 및 접종 후 혈액 채취 시간에 따른 각 그룹의 RGNNV capsid protein에 대한 혈중 항체가 측정 결과를 보여줌. RGNNV capsid에 대한 혈중 항체가는 ELISA를 통해 측정되었음. 주사용 백신의 접종량은 12.5 ㎍과 50 ㎍ 두 종류로 나누었고 백신 접종 후 혈액 채취 시간을 각각 1차 접종 후 3주, 1차 접종 후 6주, 2차 접종 후 3주로 나누었음. 대조군으로 접종 전, PBS 주사 후 3주, PBS 주사 후 6주, PBS 경구 투여 후 3주, PBS 경구 투여 후 5주 그룹으로 설정하였음
 표
넙치 모델에서 주사용, 경구용 백신 투여에 따른 중화활성 유도 확인 결과. 넙치에 주사용, 경구용 백신을 표 9 및 표 10과 같이 투여한 후 주사용 백신 2차 접종 3주후, 경구용 백신 4차 투여 3주후에 혈청을 취하고, 혈청 내 RGNNV 중화활성을 E-11 세포에 대한 감염시험을 통해 확인하였음. 각 그룹의 혈액은 각각 1/400과 1/3200로 희석하였음. Control 결과에서 ‘E-11 cell’과 ‘RGNNV’는 RGNNV를 감염시키지 않는 정상적인 세포와 RGNNV를 감염시킨 세포 상태를 각각 보여줌. Control VLP는 PBS를 주사로 투여한 그룹의 혈청 적용 결과를 보여줌. VLP 12.5와 VLP 50은 각각 RGNNV 주사용 백신 12.5 μg 투여그룹과 50 μg 투여그룹의 혈청 적용 결과를 보여줌. Control rCP는 경구백신에 대한 대조군으로 PBS를 경구로 투여한 그룹의 혈청 적용 결과를 보여줌. rCP 25와 rCP 50은 dose당 25 mg과 50 mg을 각각 4회씩 경구로 투여한 그룹의 혈청 적용 결과를 보여줌
표
넙치 모델에서 주사용, 경구용 백신 투여에 따른 중화활성 유도 확인 결과. 넙치에 주사용, 경구용 백신을 표 9 및 표 10과 같이 투여한 후 주사용 백신 2차 접종 3주후, 경구용 백신 4차 투여 3주후에 혈청을 취하고, 혈청 내 RGNNV 중화활성을 E-11 세포에 대한 감염시험을 통해 확인하였음. 각 그룹의 혈액은 각각 1/400과 1/3200로 희석하였음. Control 결과에서 ‘E-11 cell’과 ‘RGNNV’는 RGNNV를 감염시키지 않는 정상적인 세포와 RGNNV를 감염시킨 세포 상태를 각각 보여줌. Control VLP는 PBS를 주사로 투여한 그룹의 혈청 적용 결과를 보여줌. VLP 12.5와 VLP 50은 각각 RGNNV 주사용 백신 12.5 μg 투여그룹과 50 μg 투여그룹의 혈청 적용 결과를 보여줌. Control rCP는 경구백신에 대한 대조군으로 PBS를 경구로 투여한 그룹의 혈청 적용 결과를 보여줌. rCP 25와 rCP 50은 dose당 25 mg과 50 mg을 각각 4회씩 경구로 투여한 그룹의 혈청 적용 결과를 보여줌
 표
노다바이러스 주사용 백신 및 경구용 백신의 투여에 따른 능성어 혈중 항 -RGNNV capsid protein 항체 수준 측정결과. 수집한 혈액을 생리식염수에 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 skim milk로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체 -RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. control, n=10. 주사, n=10. 경구, n=10. 사료, n=10. 침지, n=10
표
노다바이러스 주사용 백신 및 경구용 백신의 투여에 따른 능성어 혈중 항 -RGNNV capsid protein 항체 수준 측정결과. 수집한 혈액을 생리식염수에 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 skim milk로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체 -RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. control, n=10. 주사, n=10. 경구, n=10. 사료, n=10. 침지, n=10
 표
노다바이러스 주사용 백신의 접종에 따른 능성어 혈 중 항-RGNNV capsid protein 항체 수준을 ELISA기법으로 측정한 결과. 수집한 혈액을 생리식염수에 1/400 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 BSA로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체 -RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 492 nm에서 OPD 발색 시약의 발색정도로 나타내었음. control, n=10. 5 μg, n=10. 10 μg, n=10. 20 μg, n=10. 40 μg, n=10. 80 μg, n=10
표
노다바이러스 주사용 백신의 접종에 따른 능성어 혈 중 항-RGNNV capsid protein 항체 수준을 ELISA기법으로 측정한 결과. 수집한 혈액을 생리식염수에 1/400 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 BSA로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체 -RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 492 nm에서 OPD 발색 시약의 발색정도로 나타내었음. control, n=10. 5 μg, n=10. 10 μg, n=10. 20 μg, n=10. 40 μg, n=10. 80 μg, n=10
 표
RGNNV 주사용 백신의 접종량에 따른 넙치 혈 중 항-RGNNV capsid protein 항체 수준을 ELISA 기법으로 측정한 결과. 수집한 혈액을 생리식염수에 three-fold로 연속 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 skim milk로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well 로 분주함으로서 어체 혈중 항체-RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. 접종 전 n=5. PBS, n=5. 10 μg, n=5. 20 μg, n=5. 40 μg, n=5. 80 μg, n=5
표
RGNNV 주사용 백신의 접종량에 따른 넙치 혈 중 항-RGNNV capsid protein 항체 수준을 ELISA 기법으로 측정한 결과. 수집한 혈액을 생리식염수에 three-fold로 연속 희석하고 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 skim milk로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well 로 분주함으로서 어체 혈중 항체-RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. 접종 전 n=5. PBS, n=5. 10 μg, n=5. 20 μg, n=5. 40 μg, n=5. 80 μg, n=5
 표
노다바이러스 주사 백신 및 경구 백신의 투여에 따른 능성어 혈 중 항-노다바이러스 중화항체 역가 측정결과. 그룹당 개별 어체 혈액을 1/50부터 1/109,350까지 3배 희석배율로 희석하여 RGNNV 바이러스와 혼합한 다음 E-11세포에 분주하였음. 세포병변이 관찰되지 않는 혈액의 최고 희석비율을 serum 중화항체 역가로 정하였음. 결과에서 그룹 당 25%-75% 값의 범위를 박스로 나타내었고 median값을 선으로 나타내었음. 접종전, n=10. 1차채혈(대조, 경구, 주사), n=10. 2차채혈(대조, 경구, 주사), n=11. 3차채혈(대조, 경구, 주사), n=12. 4차채혈(대조, 경구, 주사), n=10
표
노다바이러스 주사 백신 및 경구 백신의 투여에 따른 능성어 혈 중 항-노다바이러스 중화항체 역가 측정결과. 그룹당 개별 어체 혈액을 1/50부터 1/109,350까지 3배 희석배율로 희석하여 RGNNV 바이러스와 혼합한 다음 E-11세포에 분주하였음. 세포병변이 관찰되지 않는 혈액의 최고 희석비율을 serum 중화항체 역가로 정하였음. 결과에서 그룹 당 25%-75% 값의 범위를 박스로 나타내었고 median값을 선으로 나타내었음. 접종전, n=10. 1차채혈(대조, 경구, 주사), n=10. 2차채혈(대조, 경구, 주사), n=11. 3차채혈(대조, 경구, 주사), n=12. 4차채혈(대조, 경구, 주사), n=10
 표
RGNNV 공격시험에서 경구백신 및 주사백신의 투여에 따른 능성어 뇌 조직의 RGNNV titer 측정 결과. 능성어 마리 당 1X106 TCID50 RGNNV를 challenge하였고 virus challenge 후 2주 뒤에 뇌 조직을 회수하였음. 회수한 뇌 조직을 각각 1/200부터 1/25,600 까지 2배 배율로 희석하여 E-11세포에 분주하였음. 세포병변이 관찰되는 뇌 조직 mixture의 최고 희석 비율을 brain RGNNV titer로 정하였음. 결과는 각 그룹의 brand RGNNV titer를 log10 스케일로 설정하여 median 값으로 나타내었음. 대조, n=16. 경구, n=25. 주사, n=25
표
RGNNV 공격시험에서 경구백신 및 주사백신의 투여에 따른 능성어 뇌 조직의 RGNNV titer 측정 결과. 능성어 마리 당 1X106 TCID50 RGNNV를 challenge하였고 virus challenge 후 2주 뒤에 뇌 조직을 회수하였음. 회수한 뇌 조직을 각각 1/200부터 1/25,600 까지 2배 배율로 희석하여 E-11세포에 분주하였음. 세포병변이 관찰되는 뇌 조직 mixture의 최고 희석 비율을 brain RGNNV titer로 정하였음. 결과는 각 그룹의 brand RGNNV titer를 log10 스케일로 설정하여 median 값으로 나타내었음. 대조, n=16. 경구, n=25. 주사, n=25
 표
능성어 뇌 조직내 RGNNV titer 측정 시 E-11세포에 대한 세포병변을 현미경으로 관찰한 결과. 능성어 마리 당 1X106 TCID50 RGNNV를 challenge하였고 virus challenge 후 2주 뒤에 뇌 조직을 회수하였음. 각 그룹당 뇌 조직을 혼합하여 mixture를 만들고 mixture를 각각 1/200부터 1/25,600 까지 2배 배율로 희석하여 E-11세포에 분주하여 세포병변을 관찰하였음. 1/1,600과 1/6,400 비율에서 관찰한 현미경 결과를 대표결과로 나타내었음. 대조, n=16. 경구, n=25. 주사, n=25
표
능성어 뇌 조직내 RGNNV titer 측정 시 E-11세포에 대한 세포병변을 현미경으로 관찰한 결과. 능성어 마리 당 1X106 TCID50 RGNNV를 challenge하였고 virus challenge 후 2주 뒤에 뇌 조직을 회수하였음. 각 그룹당 뇌 조직을 혼합하여 mixture를 만들고 mixture를 각각 1/200부터 1/25,600 까지 2배 배율로 희석하여 E-11세포에 분주하여 세포병변을 관찰하였음. 1/1,600과 1/6,400 비율에서 관찰한 현미경 결과를 대표결과로 나타내었음. 대조, n=16. 경구, n=25. 주사, n=25
 표
시중 유통 넙치 brain의 RGNNV 활성도 조사 결과. 넙치 brain extract를 E-11 세포에 적용한 후 8일간 배양하고 CPE 형성 유무를 현미경 관찰을 통해 확인하였음(A). 각 culture의 세포 및 배양액 내 RGNNV capsid protein의 유무를 Western blot으로 확인하였음(B). CPE가 관찰된 배양 well은 ‘CPE’로 표기함. Groper는 106 TCID50으로 RGNNV를 능성어에 challenge한 후 brain extract를 적용한 well을 의미함. RGNNV는 E-11에 200 TCID50의 RGNNV를 직접 적용한 well을 의미함(positive control). Bar,50μm
표
시중 유통 넙치 brain의 RGNNV 활성도 조사 결과. 넙치 brain extract를 E-11 세포에 적용한 후 8일간 배양하고 CPE 형성 유무를 현미경 관찰을 통해 확인하였음(A). 각 culture의 세포 및 배양액 내 RGNNV capsid protein의 유무를 Western blot으로 확인하였음(B). CPE가 관찰된 배양 well은 ‘CPE’로 표기함. Groper는 106 TCID50으로 RGNNV를 능성어에 challenge한 후 brain extract를 적용한 well을 의미함. RGNNV는 E-11에 200 TCID50의 RGNNV를 직접 적용한 well을 의미함(positive control). Bar,50μm
 표
정제된 RGNNV VLP의 준비 용액에 따른 마우스 혈 중 항체 역가 및 중화항체 역가 측정결과. 정제된 RGNNV VLP를 각각 0.1, 0.3, 0.5, 0.8, 1.2 M NaCl을 포함한 TBST에서 투석하여 준비함. 조건별로 준비한 RGNNV VLP 항원을 Balb/c 마우스에 100 ng/dose, 2주 간격으로 총 4회 피하주사 진행하고 마지막 면역으로부터 10일 뒤 혈액을 수집하였음. 면역을 위해 별도의 면역보조제는 사용하지 않았음. 생리식염수를 주사한 마우스를 대조군으로 설정하였음. 마우스 혈 중 항-RGNNV capsid protein 항체 역가(왼쪽)는 ELISA기법으로 측정하였고 혈 중 RGNNV 중화항체 역가(오른쪽)는 E-11세포를 기반 중화항체 역가 측정법으로 측정하였음. 0.1 M NaCl, n=10. 0.3 M NaCl, n=10. 0.5 M NaCl, n=10. 0.8 M NaCl, n=10. 1.0 M NaCl, n=10
표
정제된 RGNNV VLP의 준비 용액에 따른 마우스 혈 중 항체 역가 및 중화항체 역가 측정결과. 정제된 RGNNV VLP를 각각 0.1, 0.3, 0.5, 0.8, 1.2 M NaCl을 포함한 TBST에서 투석하여 준비함. 조건별로 준비한 RGNNV VLP 항원을 Balb/c 마우스에 100 ng/dose, 2주 간격으로 총 4회 피하주사 진행하고 마지막 면역으로부터 10일 뒤 혈액을 수집하였음. 면역을 위해 별도의 면역보조제는 사용하지 않았음. 생리식염수를 주사한 마우스를 대조군으로 설정하였음. 마우스 혈 중 항-RGNNV capsid protein 항체 역가(왼쪽)는 ELISA기법으로 측정하였고 혈 중 RGNNV 중화항체 역가(오른쪽)는 E-11세포를 기반 중화항체 역가 측정법으로 측정하였음. 0.1 M NaCl, n=10. 0.3 M NaCl, n=10. 0.5 M NaCl, n=10. 0.8 M NaCl, n=10. 1.0 M NaCl, n=10
 표
경구용 및 주사용 백신 접종 능성어의 1차, 2차, 3차 채혈 혈액의 항체 역가 측정 결과. 수집한 혈액을 각각 생리식염수에 희석하여 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 BSA로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체-RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. 대조, n=10. 경구, n=10. 주사, n=10
표
경구용 및 주사용 백신 접종 능성어의 1차, 2차, 3차 채혈 혈액의 항체 역가 측정 결과. 수집한 혈액을 각각 생리식염수에 희석하여 96 well plate에 코팅하였음. Well마다 혈액이 코팅되지 않은 부분은 BSA로 blocking시켰고 이어 정제된 RGNNV VLP를 50 ng/well로 분주함으로서 어체 혈중 항체-RGNNV VLP 복합체의 결합을 유도하였음. 어체 혈중 항체-RGNNV VLP 복합체는 RGNNV capsid protein에 대한 1차 항체를 사용하여 검출하였고 최종 혈 중 항체 역가는 양성반응을 보이는 혈액의 최고 희석 비율로 나타내었음. 대조, n=10. 경구, n=10. 주사, n=10
 표
탄소원 배양 조건에 따른 주사용 백신 항원 발현 세포의 성장, 백신 항원 발현 및 항원 수율을 비교한 결과. 탄소원 6% 기준으로 D-glucose와 galactose를 각각 3:3, 4:, 5:1, 5.5:0.5 첨가하여 주사용 백신 항원 발현 효모 세포를 72 시간 동안 배양하고 A. 세포 무게를 측정함. B. 주사용 백신 항원의 발현을 Western blot으로 확인함. C. 배양한 효모 세포를 정제하여 최종 RGNNV VLP 주사용 백신 L당 수율을 확인함. D. 배양한 효모 세포를 정제하여 RGNNV VLP 주사용 백신 수율을 확인함
표
탄소원 배양 조건에 따른 주사용 백신 항원 발현 세포의 성장, 백신 항원 발현 및 항원 수율을 비교한 결과. 탄소원 6% 기준으로 D-glucose와 galactose를 각각 3:3, 4:, 5:1, 5.5:0.5 첨가하여 주사용 백신 항원 발현 효모 세포를 72 시간 동안 배양하고 A. 세포 무게를 측정함. B. 주사용 백신 항원의 발현을 Western blot으로 확인함. C. 배양한 효모 세포를 정제하여 최종 RGNNV VLP 주사용 백신 L당 수율을 확인함. D. 배양한 효모 세포를 정제하여 RGNNV VLP 주사용 백신 수율을 확인함
 표
탄소원 배양 조건 및 feeding 조건에 따른 경구용 백신 항원 발현 세포의 성장률 및 백신 항원 발현양 확인한 결과. 탄소원 6% 기준으로 D-glucose와 galactose를 각각 3:3, 4:2로 조성하여 경구용 백신 항원 발현 효모 세포를 72 - 120시간 동안 배양함. 4:2(feeding) 조건의 경우 72 시간에 galactose를 1.5% 되게 첨가하고 120시간까지 배양하였음. 4:2 조건의 경우 feeding 없이 120시간 까지 배양하였고 3:3 조건의 경우 feeding 없이 72시간 까지 배양하였음. A. 최종 배양 후 세포 무게. B. 경구용 백신 항원의 발현을 Western blot으로 확인한 결과
표
탄소원 배양 조건 및 feeding 조건에 따른 경구용 백신 항원 발현 세포의 성장률 및 백신 항원 발현양 확인한 결과. 탄소원 6% 기준으로 D-glucose와 galactose를 각각 3:3, 4:2로 조성하여 경구용 백신 항원 발현 효모 세포를 72 - 120시간 동안 배양함. 4:2(feeding) 조건의 경우 72 시간에 galactose를 1.5% 되게 첨가하고 120시간까지 배양하였음. 4:2 조건의 경우 feeding 없이 120시간 까지 배양하였고 3:3 조건의 경우 feeding 없이 72시간 까지 배양하였음. A. 최종 배양 후 세포 무게. B. 경구용 백신 항원의 발현을 Western blot으로 확인한 결과
| 과제명(ProjectTitle) : | - |
|---|---|
| 연구책임자(Manager) : | - |
| 과제기간(DetailSeriesProject) : | - |
| 총연구비 (DetailSeriesProject) : | - |
| 키워드(keyword) : | - |
| 과제수행기간(LeadAgency) : | - |
| 연구목표(Goal) : | - |
| 연구내용(Abstract) : | - |
| 기대효과(Effect) : | - |
Copyright KISTI. All Rights Reserved.

※ AI-Helper는 부적절한 답변을 할 수 있습니다.